Salt (1.5 M) avoidance test in [ChR2-XXL/Cy0 Dop1R2-RNAi/Tm3] larvae under red and blue light
on Friday, January 30th, 2026 4:23 | by Christoph Kumpfmüller
Category: crosses, Larvae, Mushroom Body, Optogenetics | No Comments
FENS abstract 2026 Ipek & Friðrik
on Monday, January 26th, 2026 12:58 | by Fridrik Kjartansson
Habit formation circuit in Drosophila Melanogaster
Fridrik Kjartansson*, Ipek Subay*, Radostina Lyutova, Björn Brembs
University of Regensburg, Zoology – Neurogenetics, Regensburg, Germany
*These authors contributed equally.
Classical learning forms association between a novel external stimulus and a
consequence of innate value. Conversely, operant learning associates animals
own behavior to such consequences; eventually habits form which ensure fast
and efficient behaviors. These forms of learning can reciprocally interact with
each other, with classical learning prioritized. A previous study from our group
demonstrated a scenario where in the case of two types of learning paradigms
introduced simultaneously in Drosophila, the classical stimulus was dominant.
However, when the training period was doubled, learning shifted to operant,
allowing habit formation in flies. Moreover, when the prominent associative
learning center, Mushroom Bodies (MB), are genetically silenced, extended
training becomes unnecessary and flies show premature habit formation.
Another study screened Mushroom Body Output Neurons (MBONs), which relay
signals from the MB and found that silencing MBON-02 specifically results in
premature habit formation suggesting MBON-02 acts as final integration center
for the selection between learning types. In this study, we utilize the power of
trans-tango and retro-tango techniques to identify the synaptic partners of
MBON-02 along with validating our results with existing connectomics datasets to
map the whole circuit that regulates the switch between these two types of
associative learning. Through behavioral testing using tethered flight, we will also
narrow down which types of MB cells, called Kenyon cells (KC) , are necessary for
the dominance of classical stimuli in such trials; furthermore, we’ll target
dopaminergic PAM neurons which form feedback loops with MBONs and KC to
elucidate their involvement.
Category: Habit formation, Operant learning, retro-tango, trans-tango | No Comments
abstract
on Monday, January 26th, 2026 12:51 | by Julia Schulz

Category: Uncategorized | No Comments
Retro-tango confocal images and Torquemeter practice results
on Monday, January 26th, 2026 12:43 | by Ipek Subay
Retro-tango:
(2023) retro-Tango enables versatile retrograde circuit tracing in Drosophila eLife 12:e85041. https://doi.org/10.7554/eLife.85041
Retro-tango genotype: y[1] w[*] P{y[+t7.7] w[+mC]=QUAS-mtdTomato-3xHA.S}su(Hw)attP8; P{y[+t7.7] w[+mC]=retro-Tango(panneuronal)}attP40/SM6b; P{y[+t7.7] w[+mC]=10xUAS-retro-Tango(ligand)-P2A-EGFP-F}attP2
Crossed with IS69306 (BDSC 601295 and 75552) split-Gal4 driver to express in b1 motor neurons. CNSs stained with anti-GFP Rabbit, (2nd ab: goat anti-rabbit 488), anti-HA Rat (2nd ab: goat anti-rat 555), nc82 (2nd ab: goat anti-mouse 647)
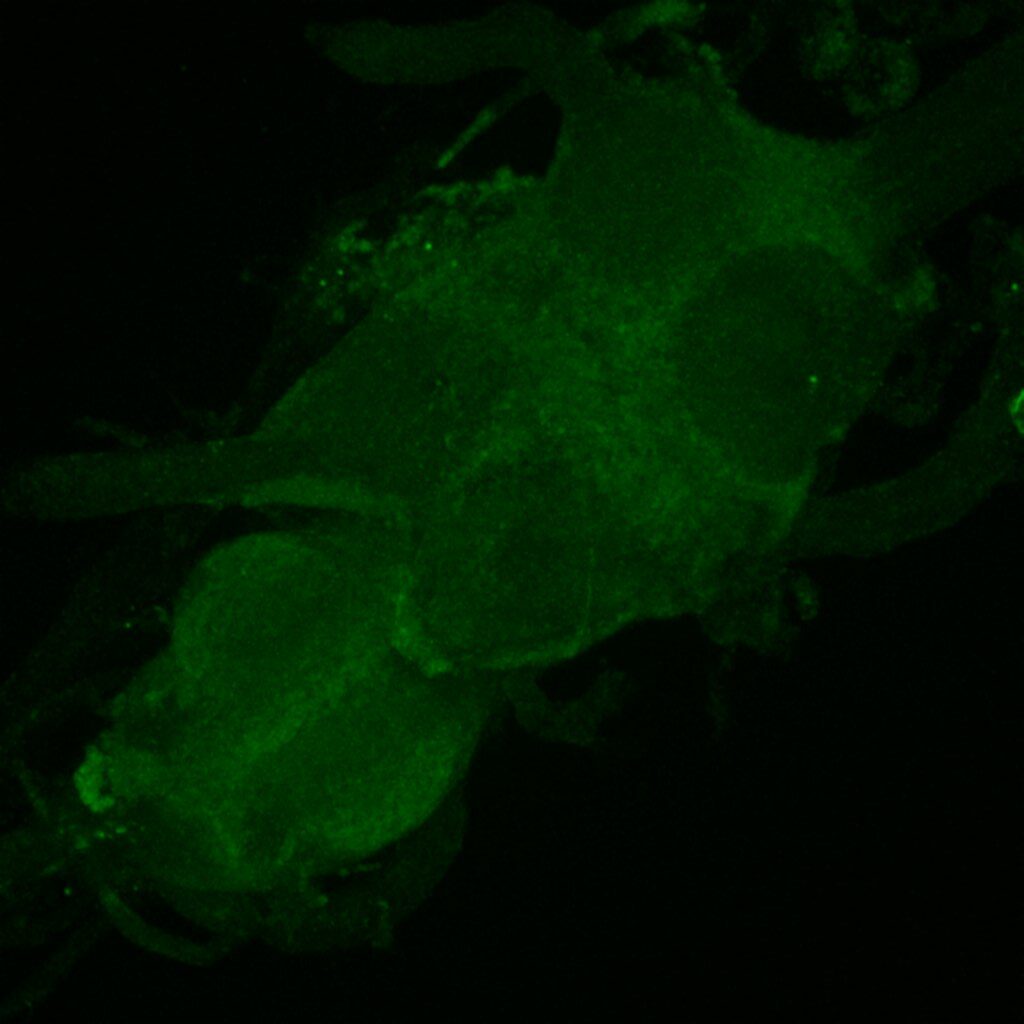
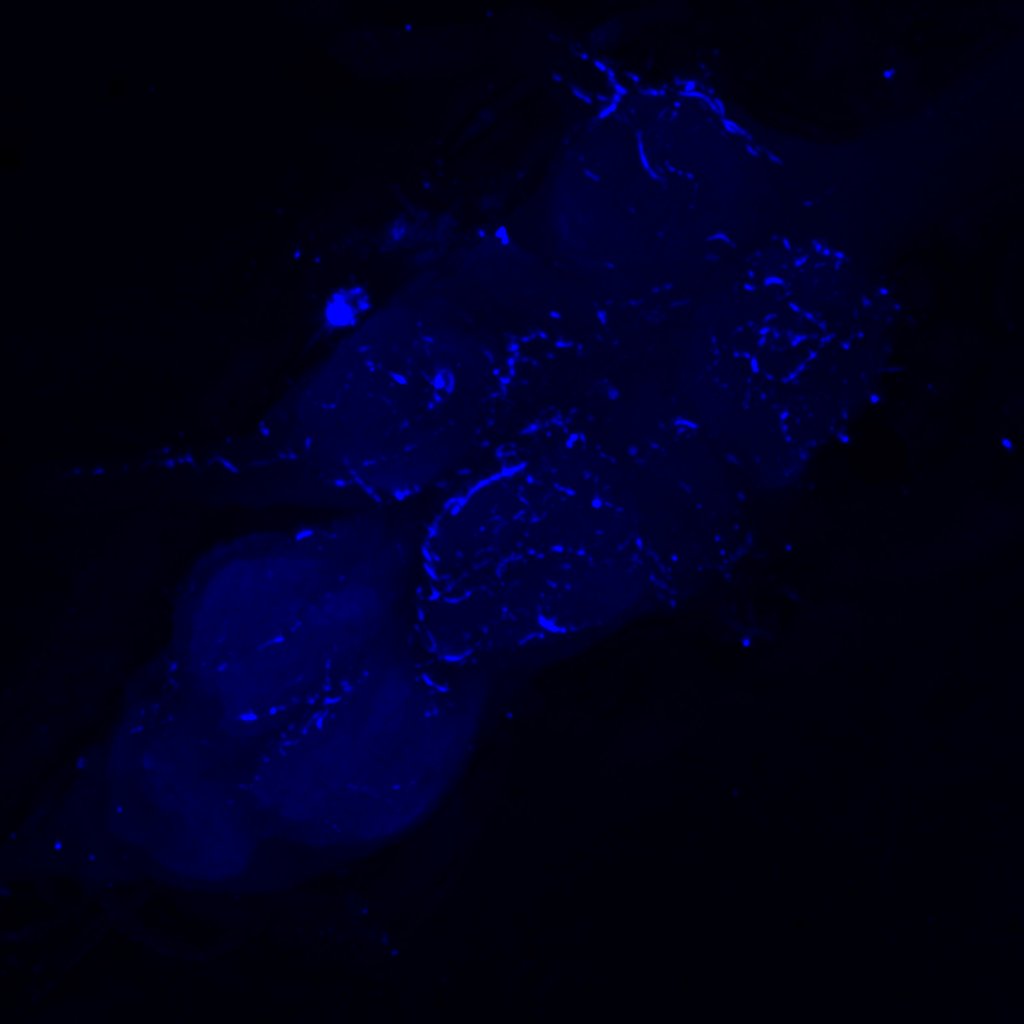
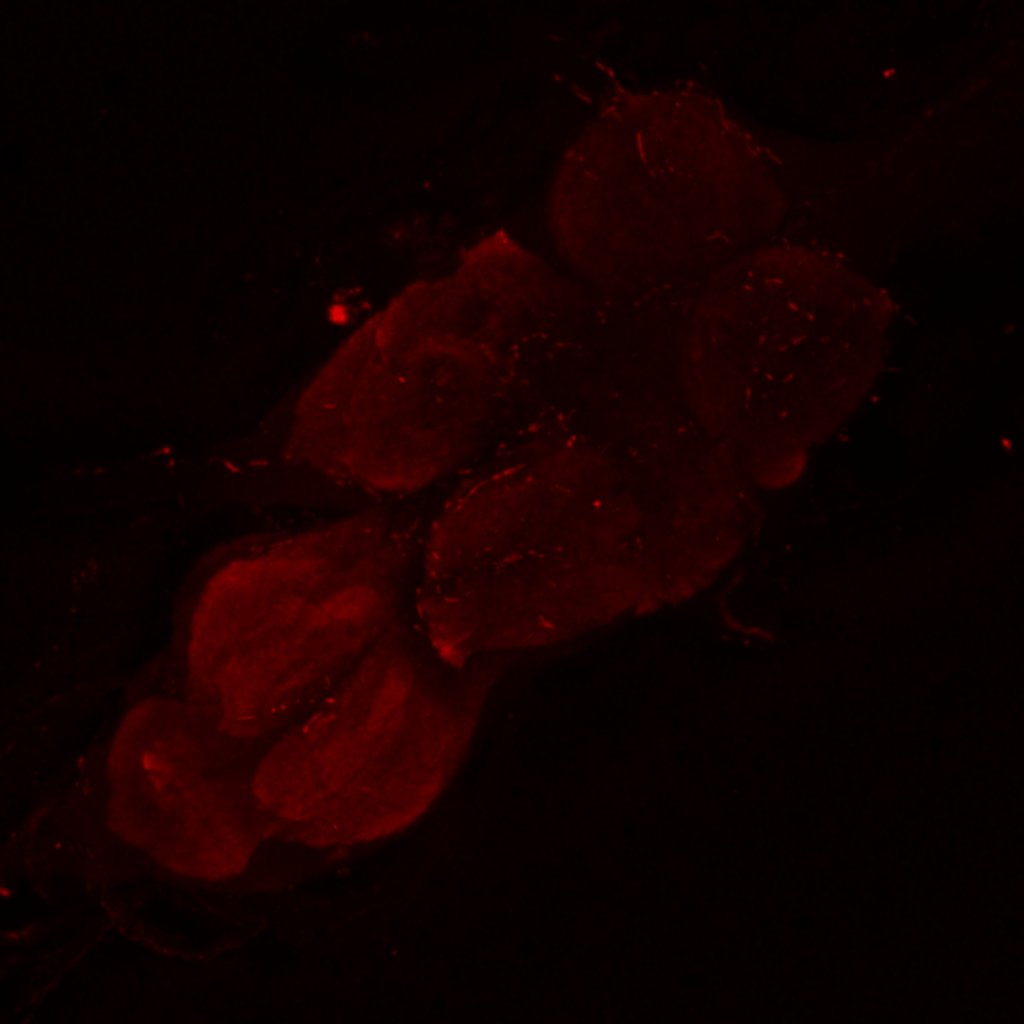
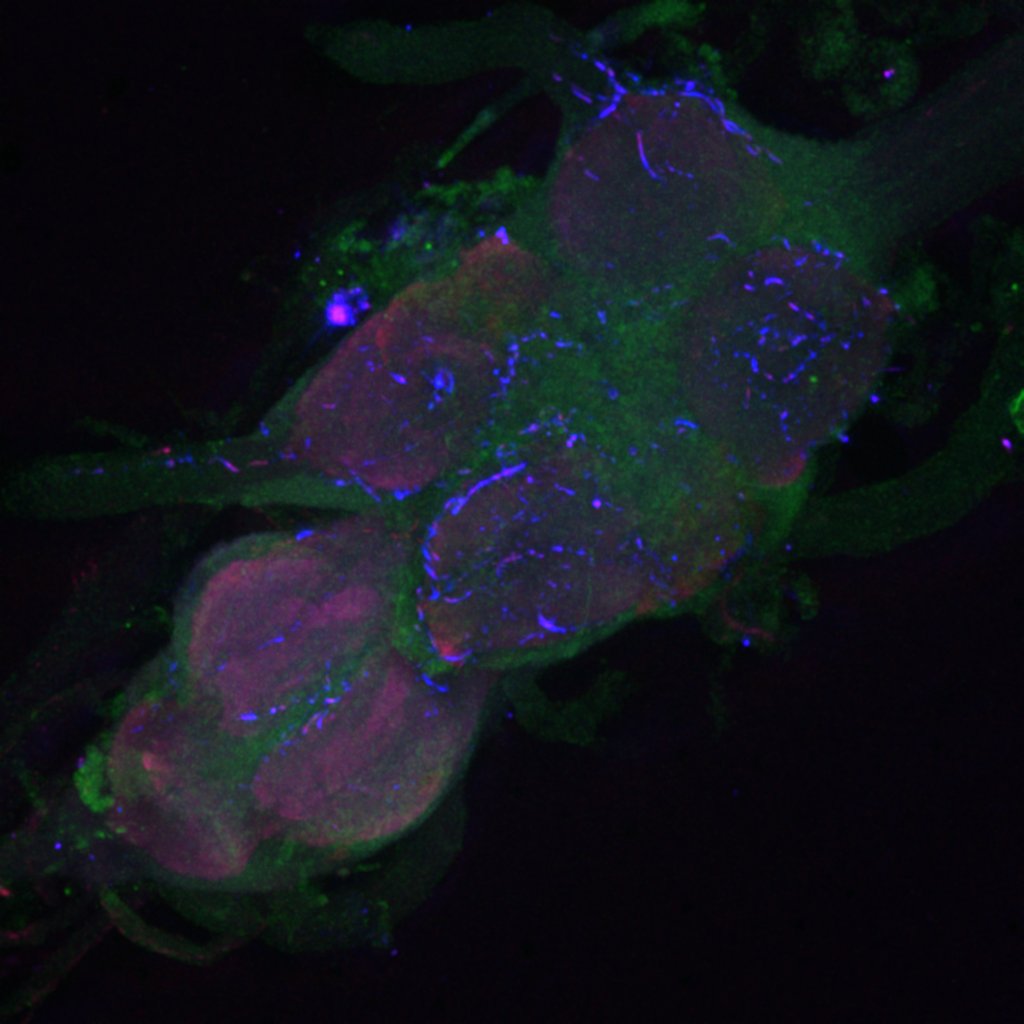
Channel 1 (Green: Laser Line ( 496 nm) Intensity: 23.99%) Spectral Positions/Gain/Offset: (501nm – 556nm) / 865.7 / -0.03
Channel 2 (Blue: Laser Line ( 561 nm) Intensity: 13.10%) Spectral Positions/Gain/Offset: (566nm – 639nm) / 537.7 / 0.03
Channel 3 (Red: Laser Line ( 633 nm) Intensity: 23.99%) Spectral Positions/Gain/Offset: (644nm – 776nm) / 802.9 / -0.01
Second line retro-tango crossed with SS98650 (split-Gal4 driver line from Janelia targeting b3 motor neurons)
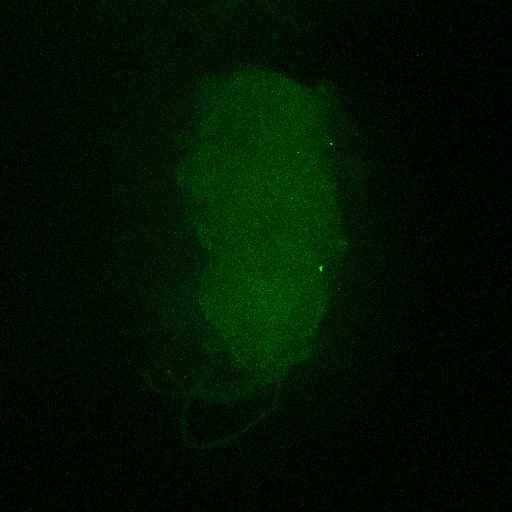
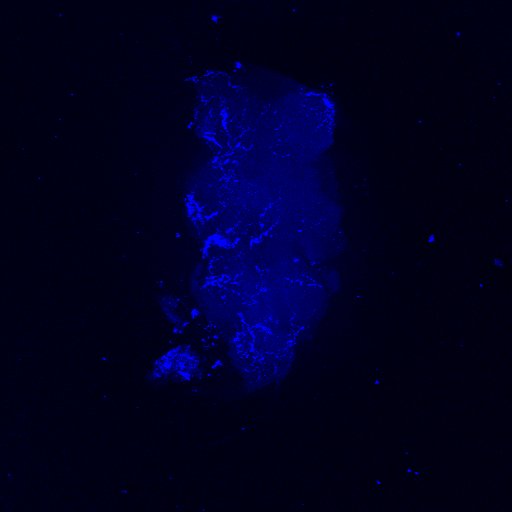
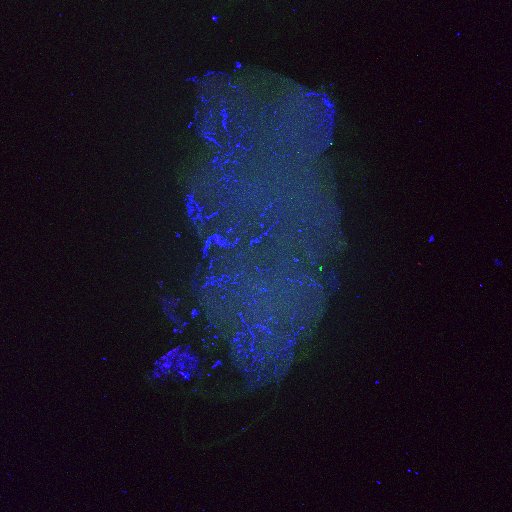
Settings are same as above
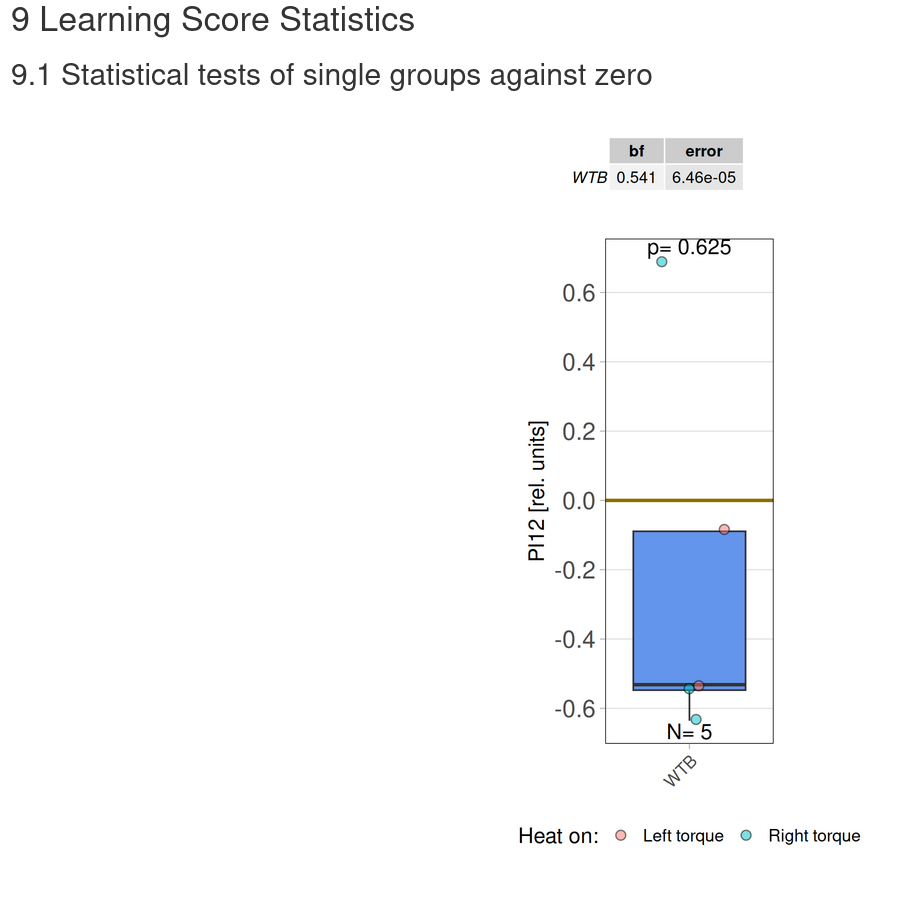
Torquemeter Practice with WTB Flies
N=10 out of 24 glued flies
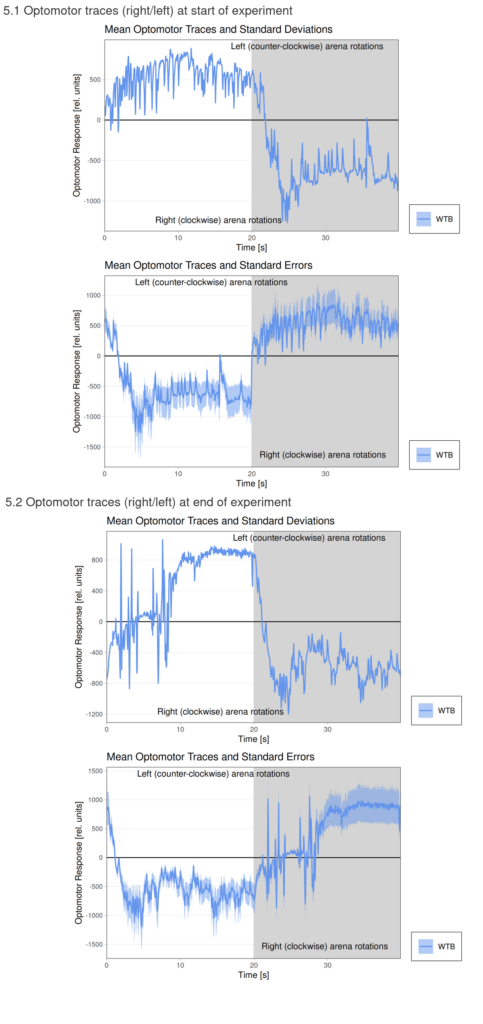
Optomotor at start:
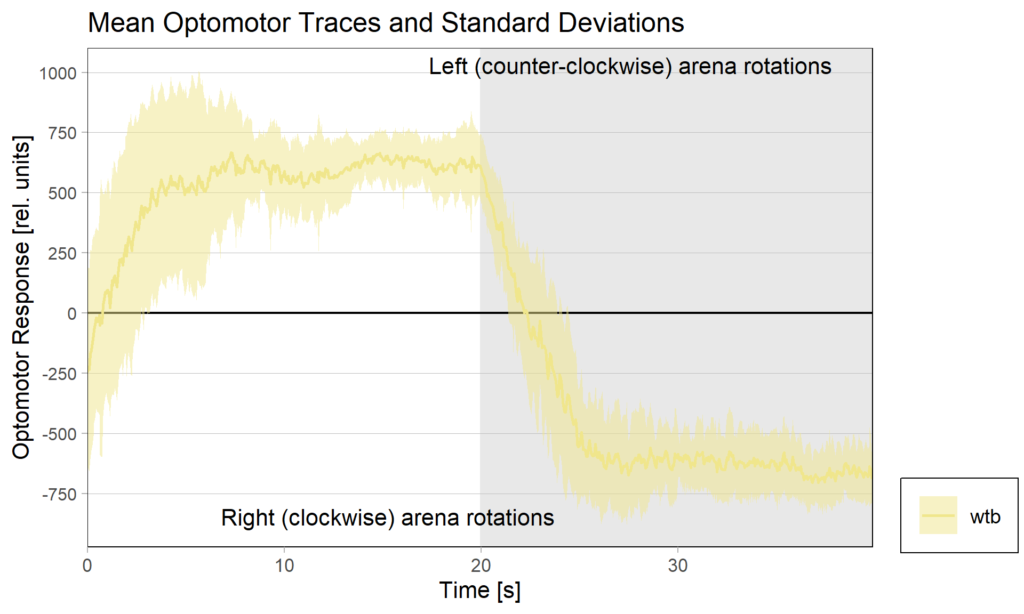
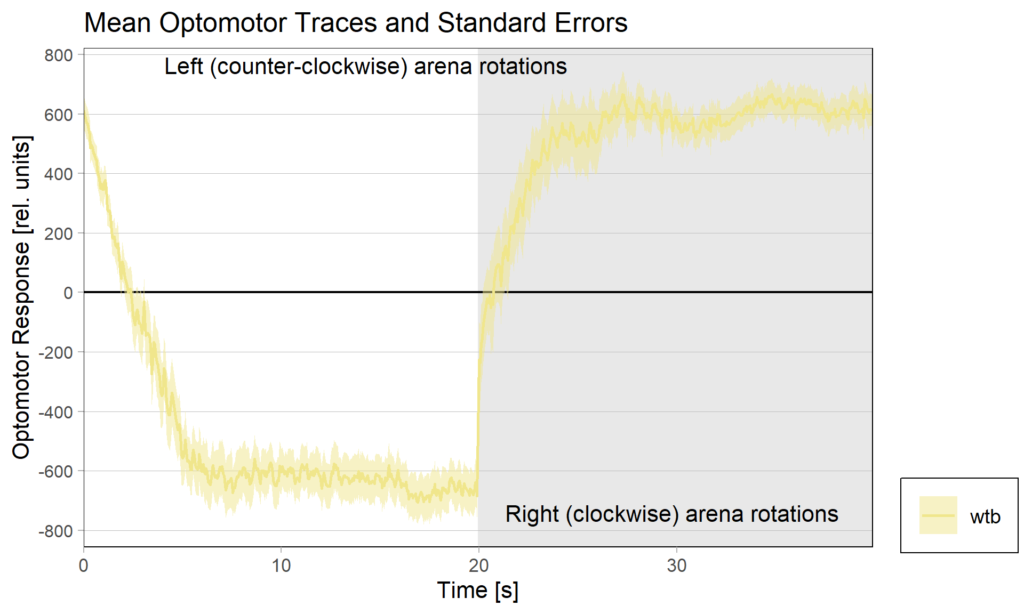
Optomotor end:
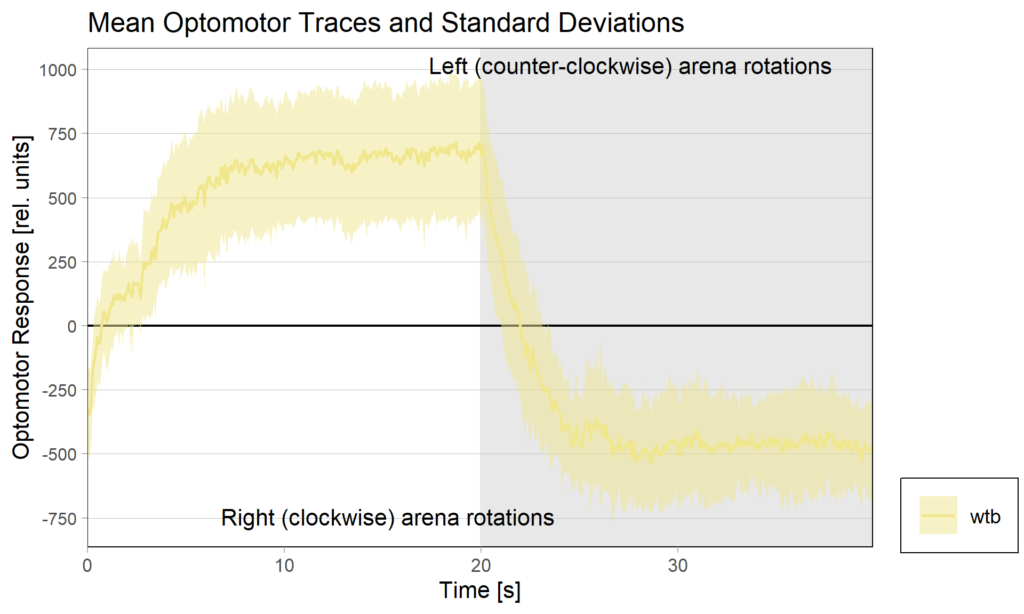
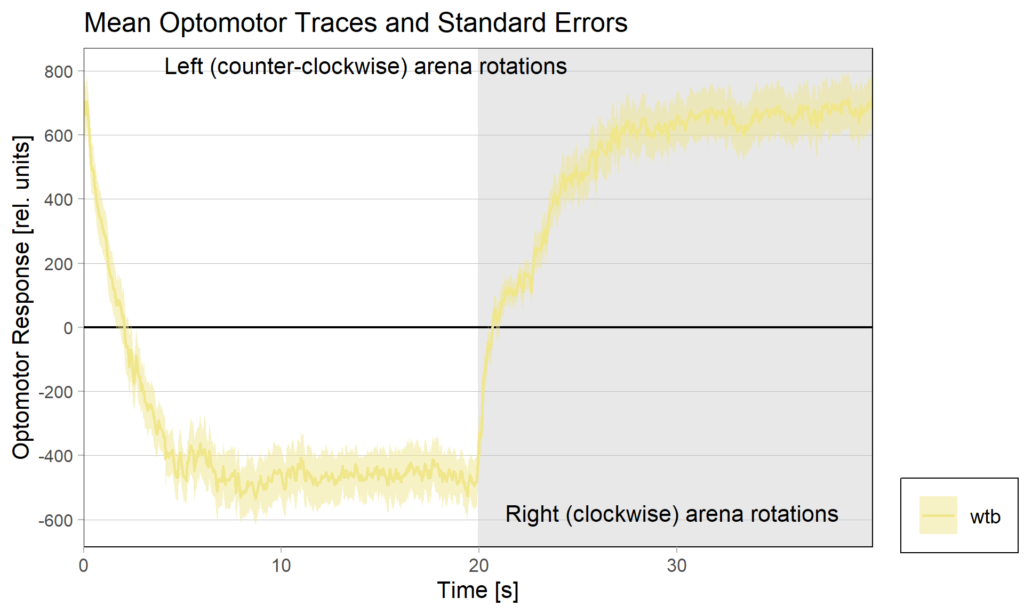
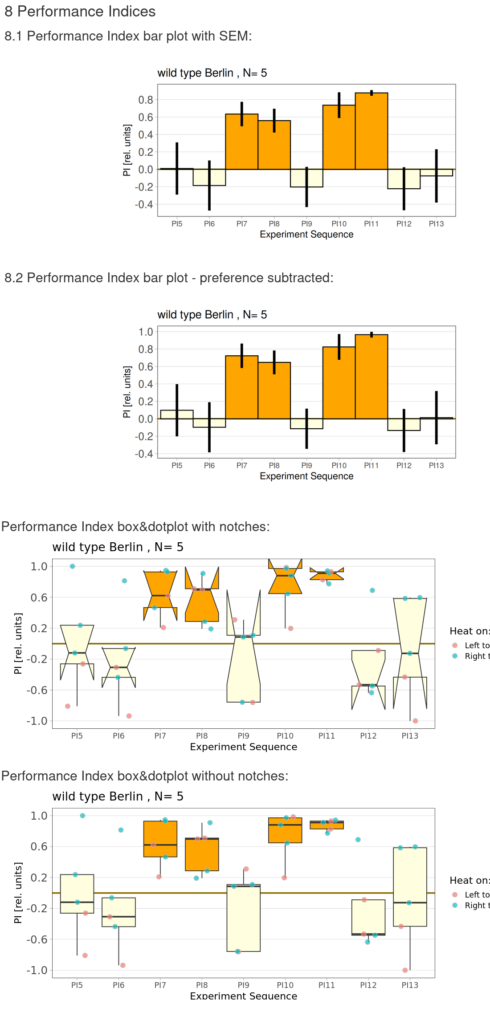
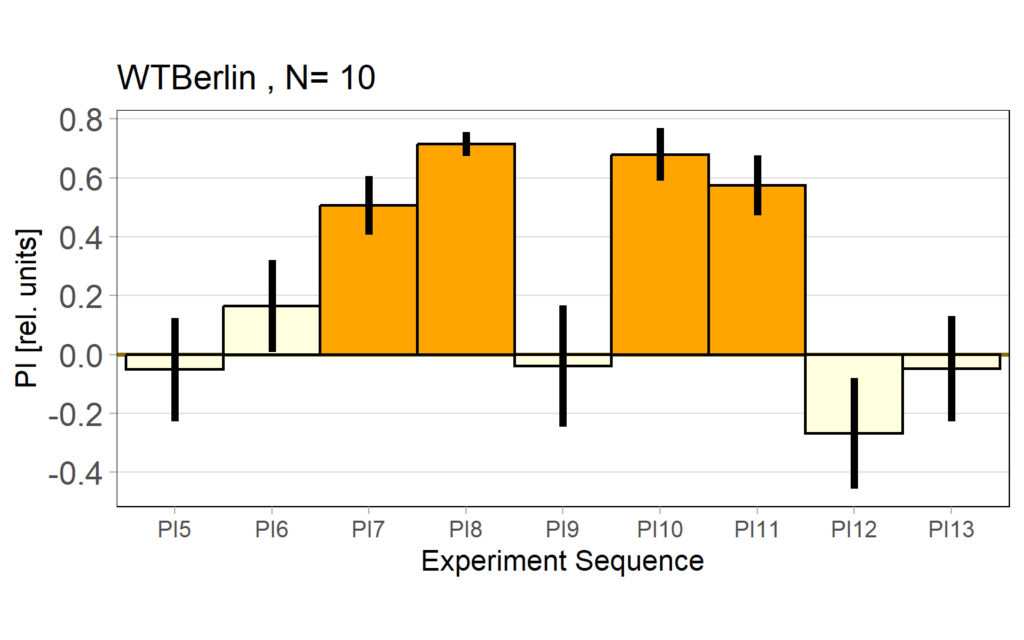
Performance index:
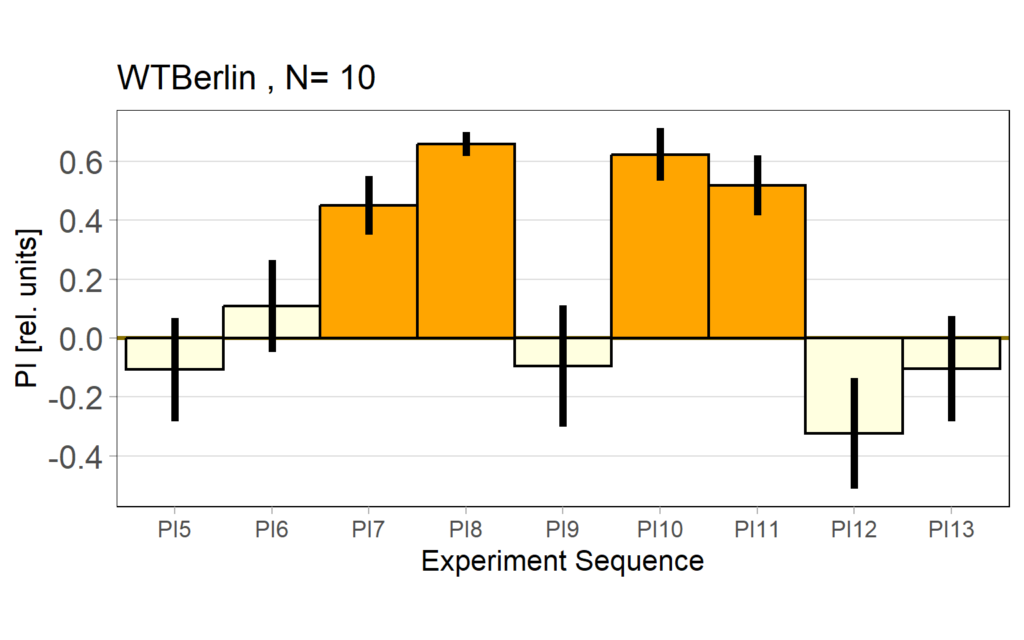
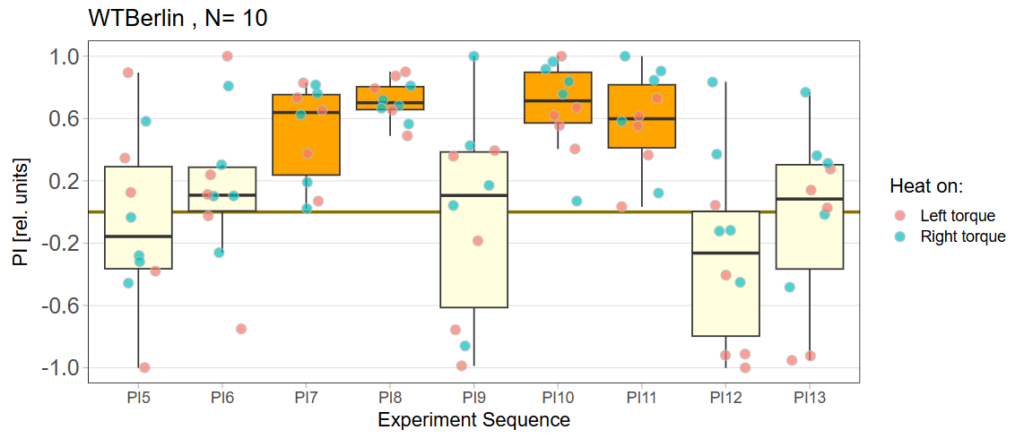
Performance subtracted:
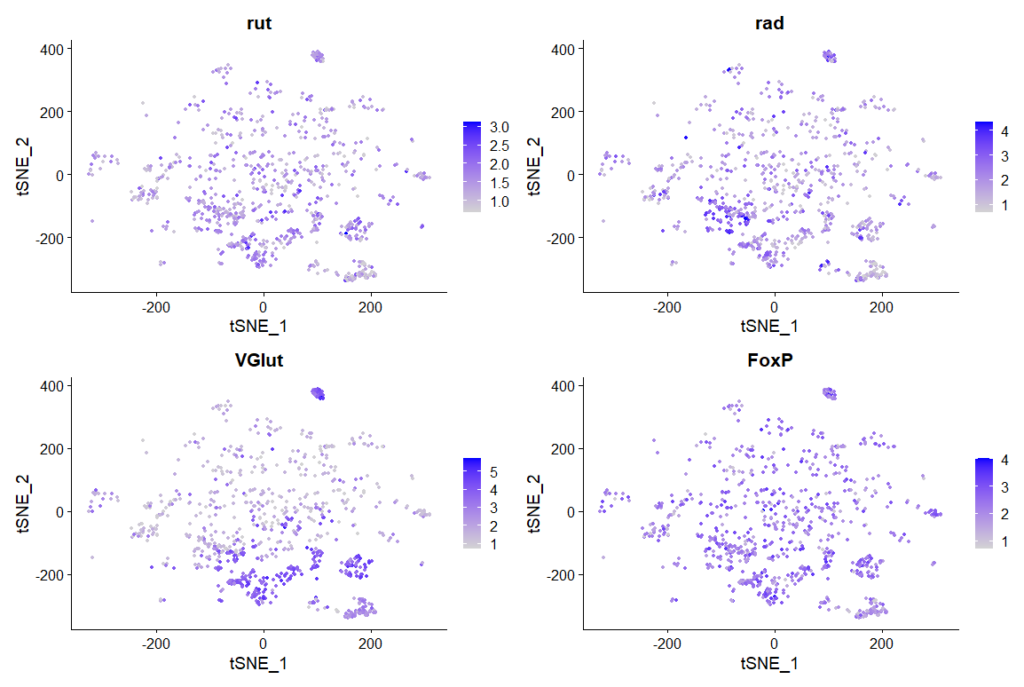
rut and rad expression in ventral nerve cord:
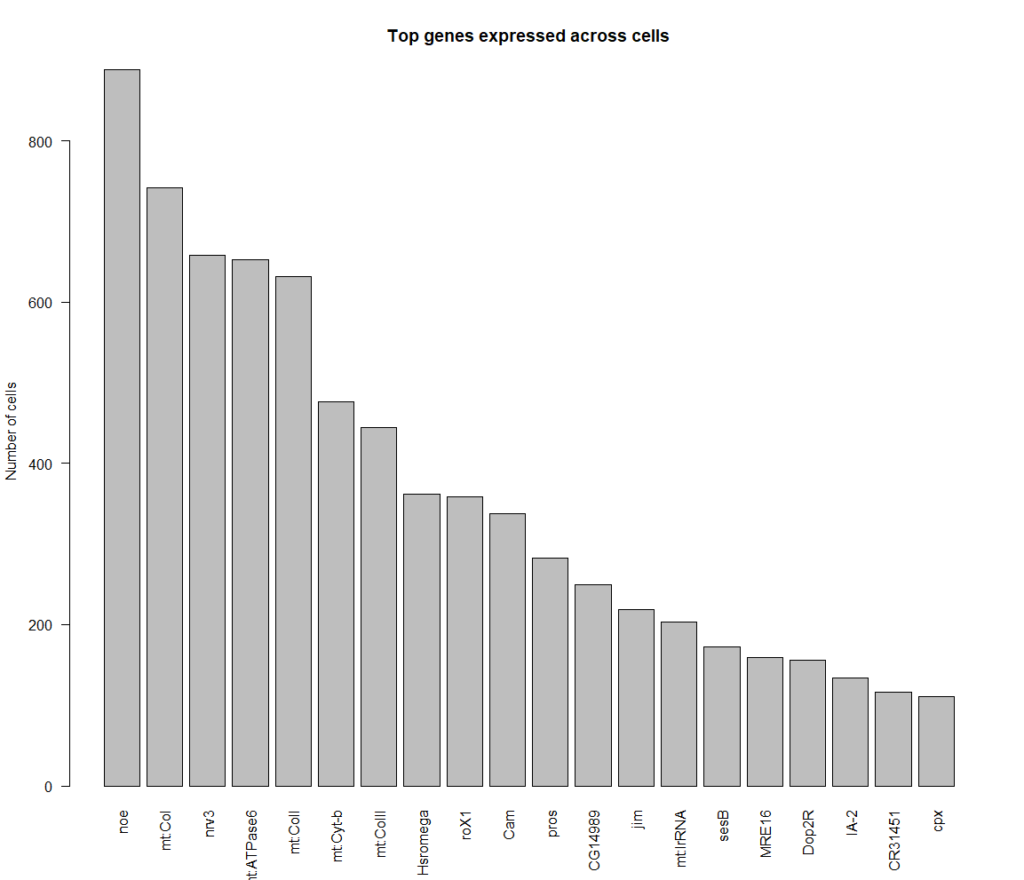
cells that express all 4 genes more than 2 fold, 889 cells in total
Top genes expressed in all of these cells:
Category: Habit formation, retro-tango | No Comments
Flexible Valence Coding by Dopaminergic PPM2 Neurons in Drosophila
on Monday, January 26th, 2026 12:11 | by Daniel Döringer
Dopaminergic systems are involved in various physiological processes including motivation and valuation. Studies on Drosophila melanogaster imply that certain insect dopaminergic neurons (DANs) are central for the mediation of valuation, as activity in these neurons can substitute for teaching signals in classical conditioning. This view may oversimplify the complex circuitry of the fly’s dopaminergic system. We focus on an understudied population of DANs and their involvement in valence coding. Using a combination of optogenetics and different operant self-stimulating paradigms, we investigated whether flies expressing an optogenetic channel in DANs of the posterior protocerebral posterior medial cluster (PPM2) would approach or avoid optogenetic stimulation. Flies’ choice was not consistent over the course of our experiments: Initially, animals avoided the stimulating light but this avoidance weakened over time and shifted to mild approach behavior in the final stages. Pharmacologically impairing DA synthesis abolished both effects. Actively exposing flies to the stimulating light aimed to identify whether the valence shift was due to accumulating neural signaling/dopamine release during the experiments. However, exposing flies to light before testing did not induce any preference changes in one-minute choice, hinting that the behavioral change does not occur from prolonged signaling in these neurons. The observation that the very same dopaminergic neurons can mediate both, avoidance and approach behavior in our self stimulating experiments, challenges a central, dopamine-mediated valuation system. Additional findings from flies that express the optogenetic channel in heat-sensing neurons further weaken the claim of centralized dopaminergic punishment neurons, as heat-punishment was independent of dopamine.
Category: Optogenetics | No Comments
Optogenetic activation of Mushroom body Kenyon cells affects naïve salt avoidance in Drosophila larvae
on Monday, January 26th, 2026 11:58 | by Radostina Lyutova
The mushroom bodies (MBs) are known to be the most prominent olfactory memory center in Drosophila. The MB Kenyon cells receive olfactory (via projection neurons, PNs) and gustatory (via dopaminergic neurons, DANs) input. The coincident detection of these stimuli elicits then the formation of associative memory. However, little is known whether and how this high order brain center affects naïve behaviors such as olfaction and gustation. We use fruit fly larvae to test the innate salt avoidance in a simple choice assay. Drosophila larvae are tested for 3 minutes for their preference for 1.5M NaCl. We show that salt avoidance is enhanced upon optogenetic activation of the Kenyon cells. This effect was also observed after ablation of pPAM DANs with simultaneous optogenetic activation of the KCs. We were able to phenocopy this effect by optogenetic activation of TH-Gal4 positive dopaminergic neurons, however not after artificial activation of single DANs of the DL1 cluster. In contrast, larvae showed no salt avoidance after optogenetic activation of sNPF-Gal4 positive neurons. Neuropeptidergic and dopaminergic signaling are tightly involved in modulation of the MBs. However, it is not clear whether our observations depend on coherent neuronal pathways. Therefore, we use simultaneous manipulations of different components of the MB, dopaminergic and sNPF circuitries to unravel the neural mechanisms of modulation of naïve gustatory behaviors in the Drosophila larvae.
Category: Biogenic Amines, DAN, Food preference, Kenyon cells, Larvae, Mushroom Body, neuronal activation, Optogenetics, PAM | No Comments
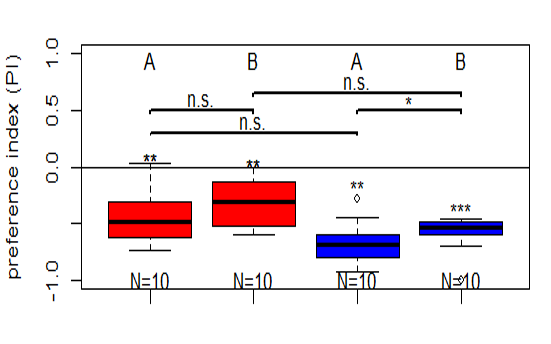
Salt avoidance test in control larvae (WTB) under red light and blue light (combined data)
on Monday, January 26th, 2026 10:35 | by Lucia Grünberger
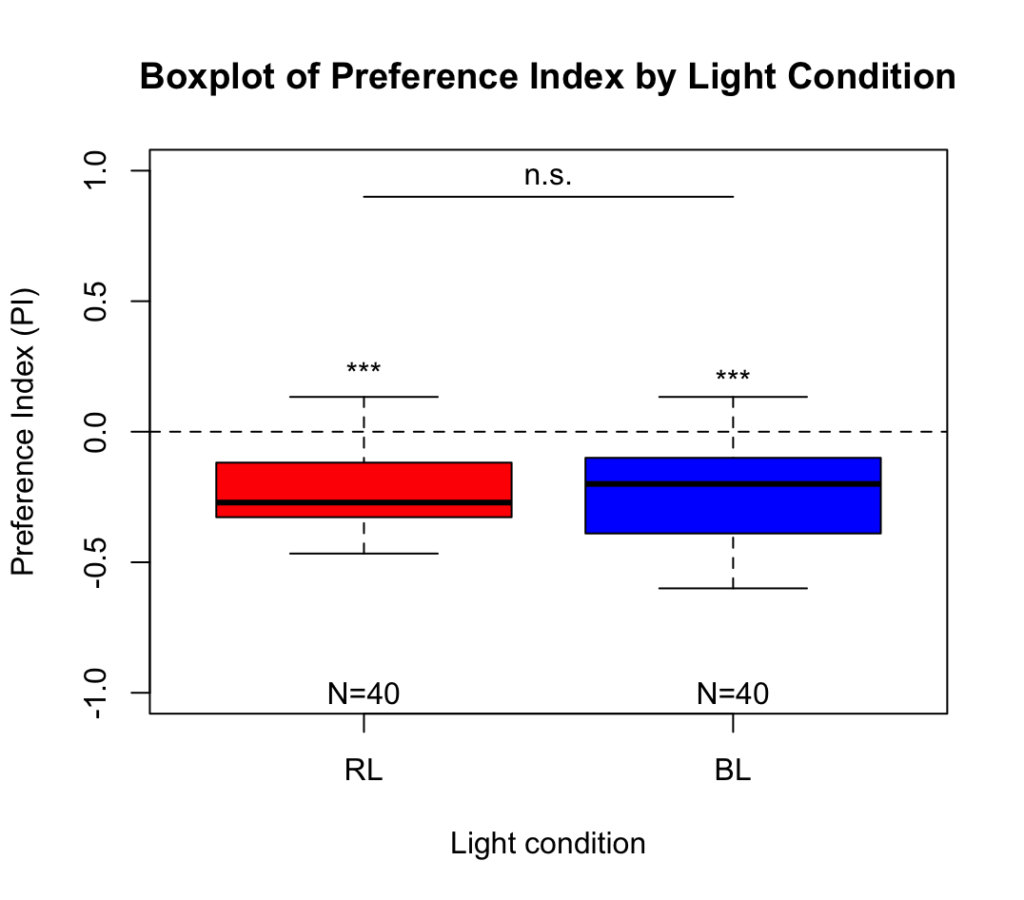
Larvae showed a significant avoidance of salt under both light conditions. When data from both weeks were combined, no significant difference in avoidance was observed between blue and red light.
Category: Food preference, Larvae | No Comments
Salt avoidance test in control larvae (WTB) under red light and blue light
on Monday, January 26th, 2026 10:26 | by Lucia Grünberger
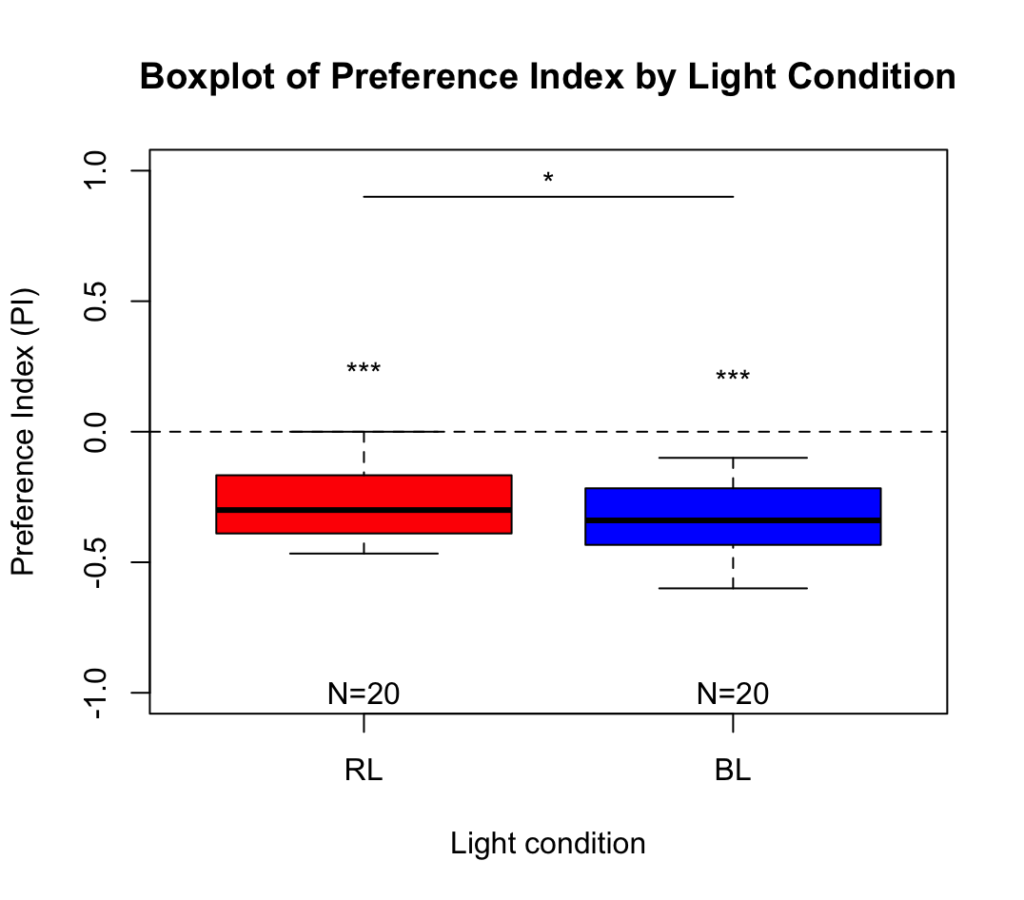
WTB Drosophila larvae were placed in the center of an agar plate with plain agar on one side and salt-containing agar on the other. After 3 minutes, larval distribution was quantified as a preference index under red and blue light.
Larvae showed a significant avoidance of salt under both light conditions. Avoidance was significantly stronger under blue light than under red light.
Category: Food preference, Larvae | No Comments
aPKC/FoxP colocalization confocal images
on Friday, January 23rd, 2026 12:52 | by Fridrik Kjartansson
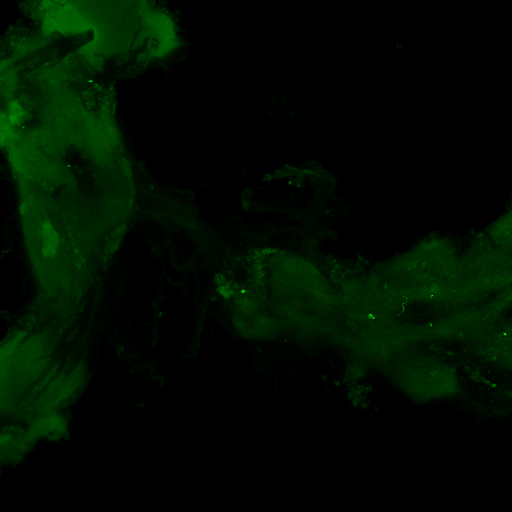
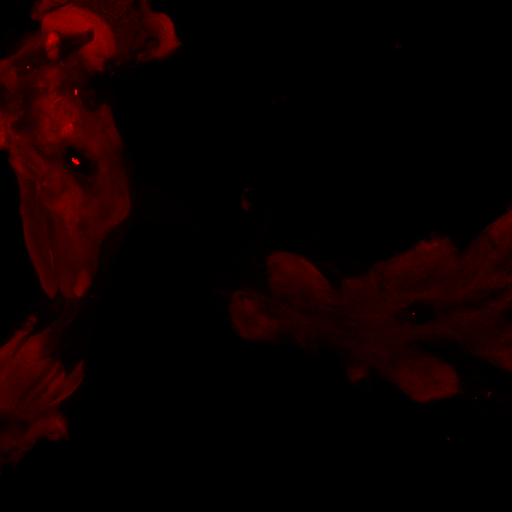
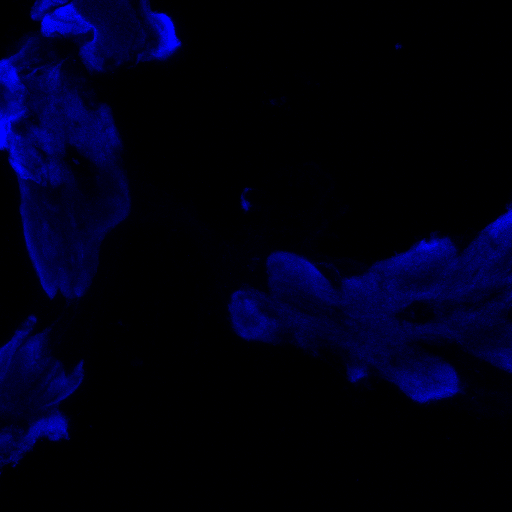
Staining is a bit faint and brain quality a bit sub-optimal and no punctae can be observed in the FoxP channel, decreasing the gain did not resolve this, blocking was done for 1 hour at RT. Perhaps increasing concentration of 1st degree antibodies, especially for RFP might improve the results.
Category: Anatomy, crosses, Foxp, PKC, PKC_localisation | No Comments