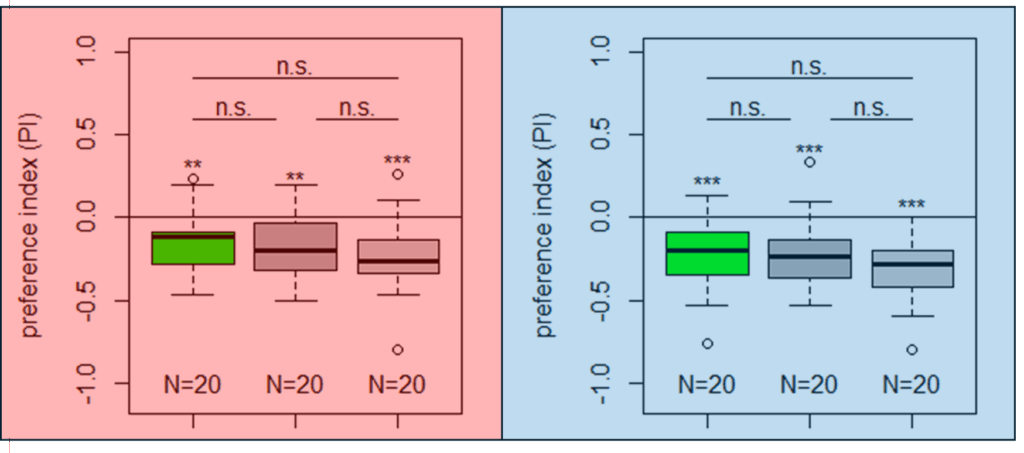
Salt (1.5 M) avoidance test in TH-D1-Gal4 larvae under red and blue light
on Saturday, November 15th, 2025 2:53 | by Christoph Kumpfmüller
Category: DAN, Larve, MBON, Mushroom Body, Optogenetics | No Comments
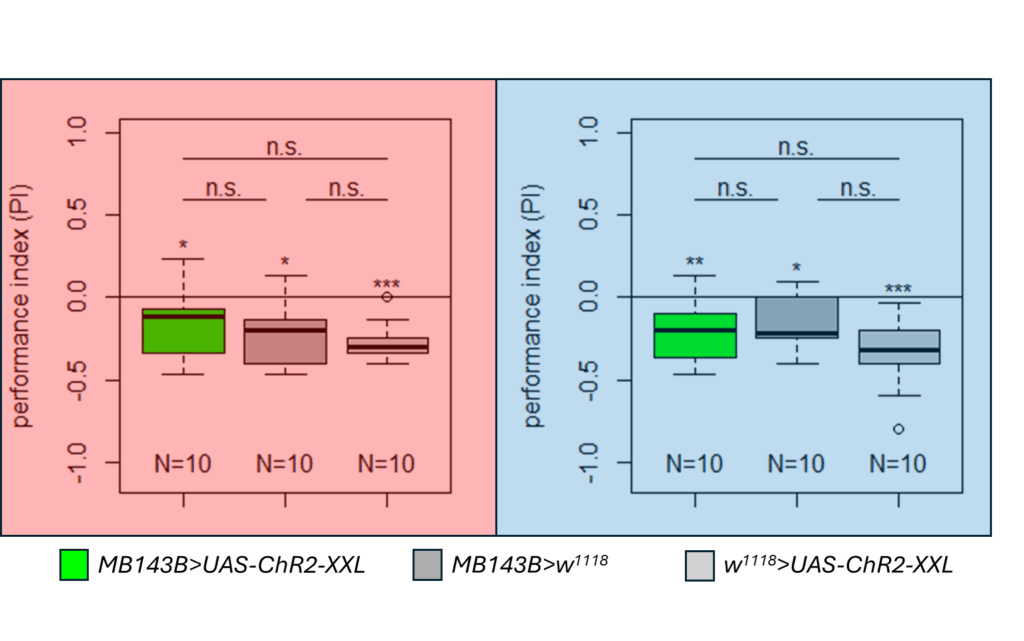
Finalized Results for the Salt (1.5 M) avoidance tests in DANc1 larvae under red and blue light
on Saturday, November 15th, 2025 2:44 | by Christoph Kumpfmüller
Category: crosses, DAN, Larve, Mushroom Body, Optogenetics | No Comments
Yellow T-Maze Results
on Friday, November 14th, 2025 3:35 | by Daniel Döringer
I performed the first set of T-Maze experiments, which included 3IY treated flies, with red light. In this experiment I could nicely reproduce earlier results in PPM2 flies only treated with ATR. Flies initially showed weak avoidance of optogenetic stimulation, and developed a weak approach-behavior over the time course of ten minutes. In the 3IY treated flies I found similar avoidance after 1 minute of testing but, interestingly, flies kept the same level of avoidance also for a choice time of 10 minutes. This indicates that initial avoidance might be independent of dopamine, but prolonged or repeated release of the neurotransmitter from PPM2 neurons might lead to circuit changes, weakening avoidance behavior, potentially even changing it to approach.
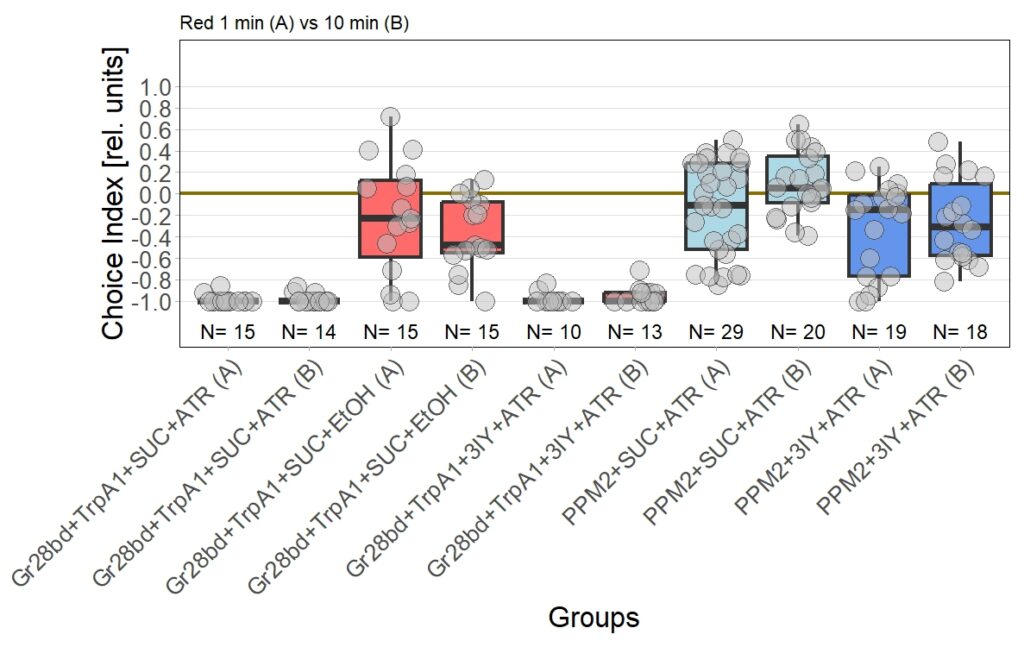
The new set of experiments uses yellow light instead of red light. This experiments are especially interesting, as I observed stronger effects for the experimental group for yellow light.
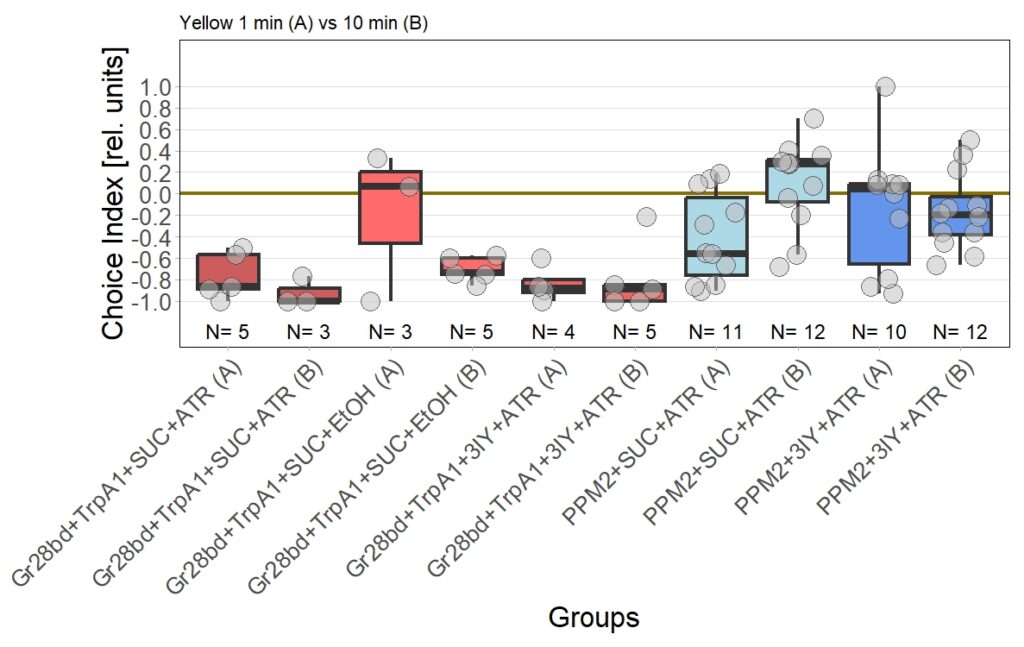
There are two main points to discuss about the data.
First, the negative control (Gr28bd+SUC+EtOH) … I was hoping that I solved the problem with the avoidance in flies that were not treated with ATR. These flies should not avoid optogenetic stimulation since without the chromophore, there should be no, or at least very weak, activation of the CsChrimson channel. Even if the sample size of 5 is rather low, it is a bit worrying that when these flies were tested for 10 minutes ((Gr28bd+SUC+EtOH (B)), they show avoidance comparable to control flies that were treated with ATR and tested for 1 minute ((Gr28bd+SUC/3IY+ATR (A)).
On the other hand, the experimental groups look pretty good. For now, I was not only able to reproduce results from the first 1 vs. 10 minute T-Maze testing with yellow light, it also seems that 3IY-treated PPM2 flies show the same phenotype as when tested in red light.
For the next few weeks I will have to increase sample sizes, aiming for 30 for each of the experimental groups. I will only include a few control groups treated with ATR, as the effect size here seems to allow for a lower N. Presumably, I will include more untreated control flies, to see whether the avoidance will persist or if the current results simply arise from the low sample size.
Update 28/11/25
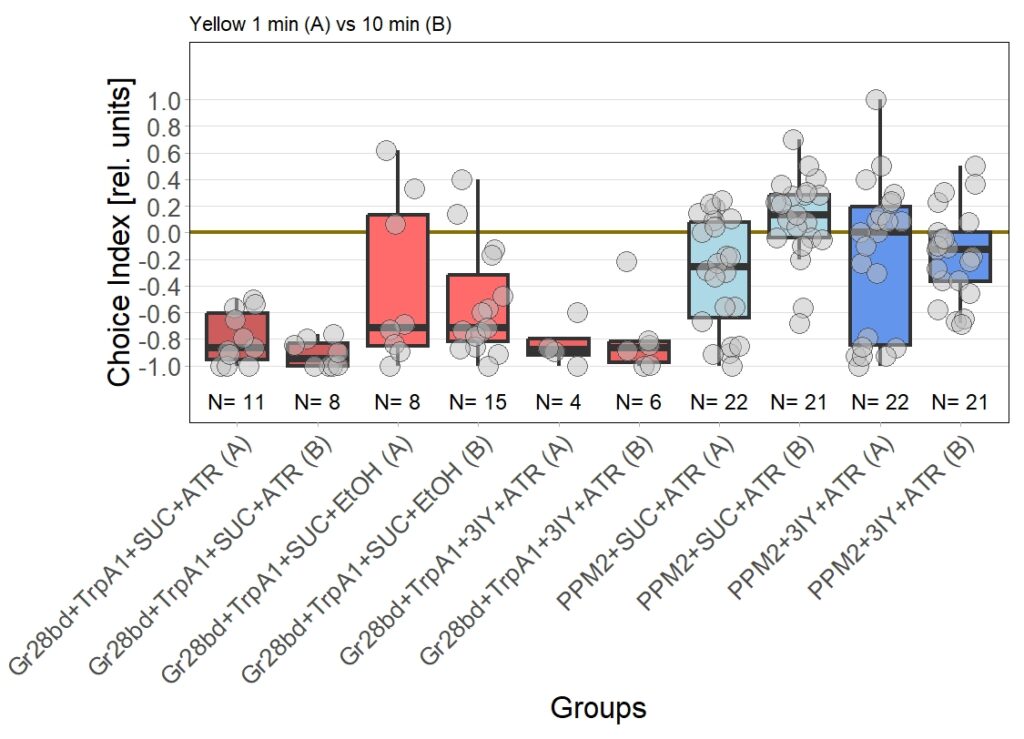
Category: Biogenic Amines, Optogenetics | No Comments
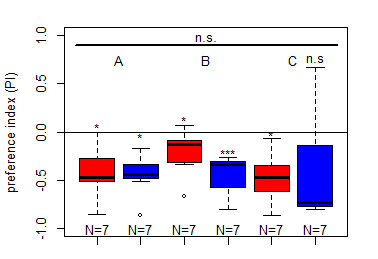
Update to the Salt (1.5 M) avoidance tests for the DANc1 larvae tests under red and blue light
on Sunday, November 9th, 2025 5:25 | by Christoph Kumpfmüller
Category: crosses, DAN, Larve, Mushroom Body, Optogenetics | No Comments
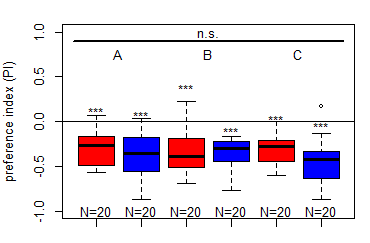
First results for Salt (1.5 M) avoidance tests in group A, B and C under red and blue light
on Sunday, November 2nd, 2025 5:27 | by Christoph Kumpfmüller
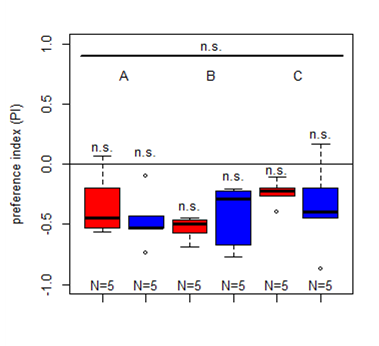
Category: crosses, DAN, Larve, Mushroom Body, Optogenetics | No Comments
Proceeding with T-Maze experiments
on Friday, October 31st, 2025 1:38 | by Daniel Döringer
After pausing T-Maze experiments because of the issues with my negative control, I should now be abled to proceed.
Since it’s been a bit more than 2 months since the last results, this was the state of the experiments back then:
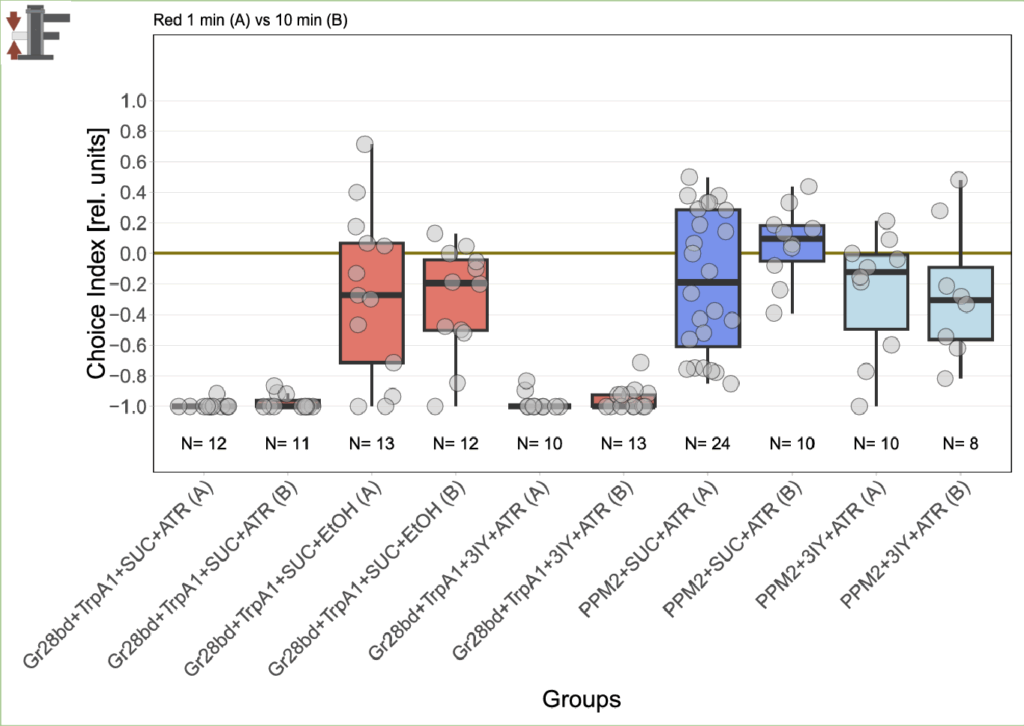
Now I added the following results:
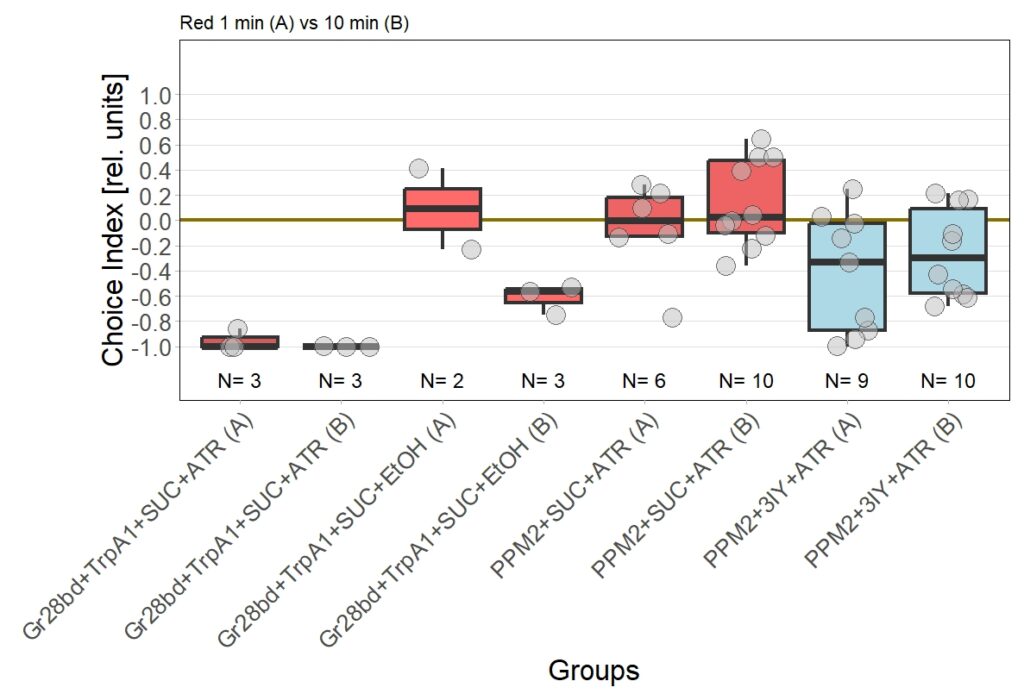
Leading to an updated version of all results:
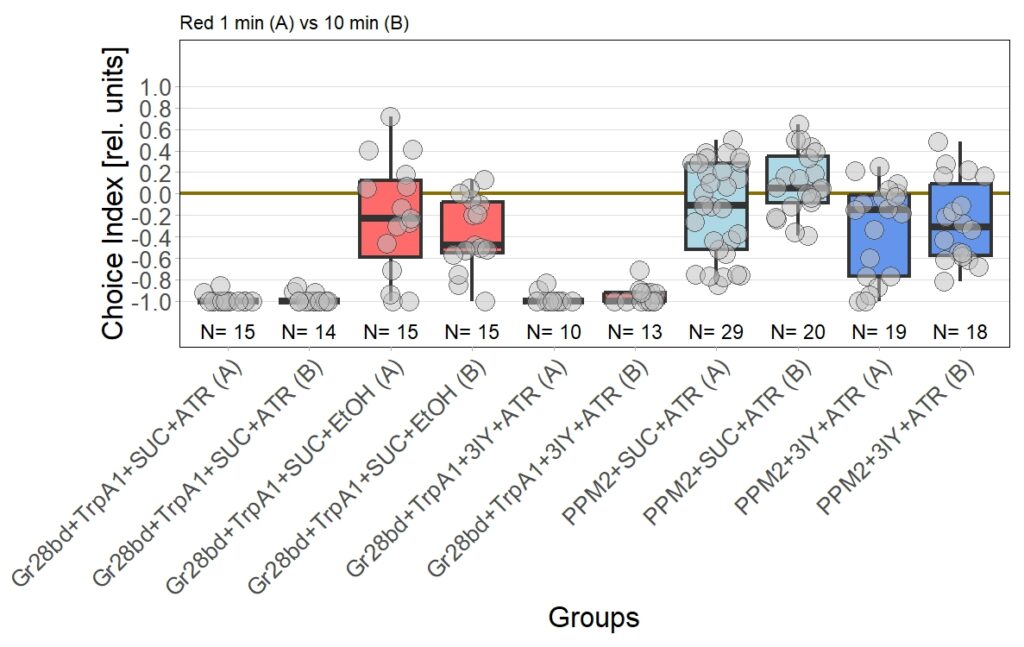
Category: Optogenetics | No Comments
Almost there
on Monday, October 27th, 2025 12:47 | by Daniel Döringer
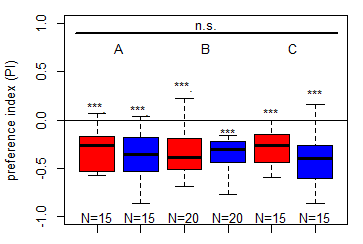
In this set of JoyStick experiments, I tested flies expressing the CsChrimson channel in OA-VuMa octopaminergic neurons, which are the Drosophila counterparts to the honeybee OA-VUMmx1 neurons. There are still a few flies left, but for now activation of these neurons does not seem to mediate any strong innate valence. There may be a slight tendency for approach in the alter periods but it might also be by chance, since we can already observe positive values in the pretest.
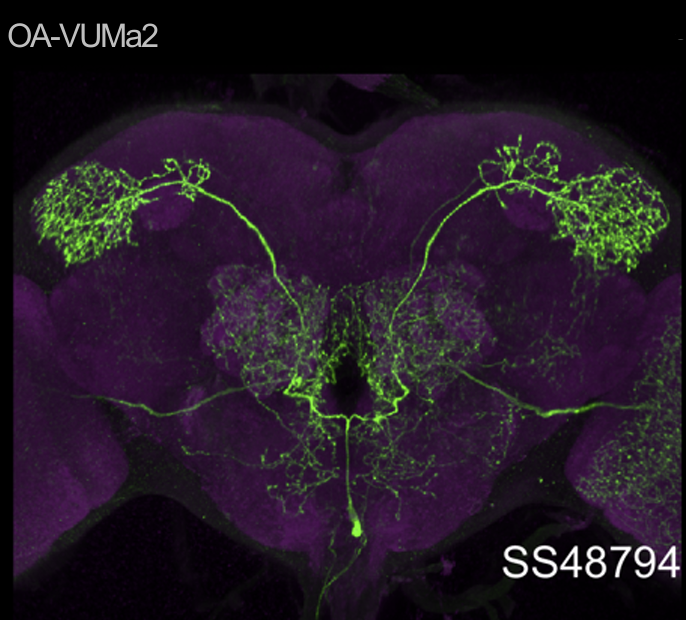
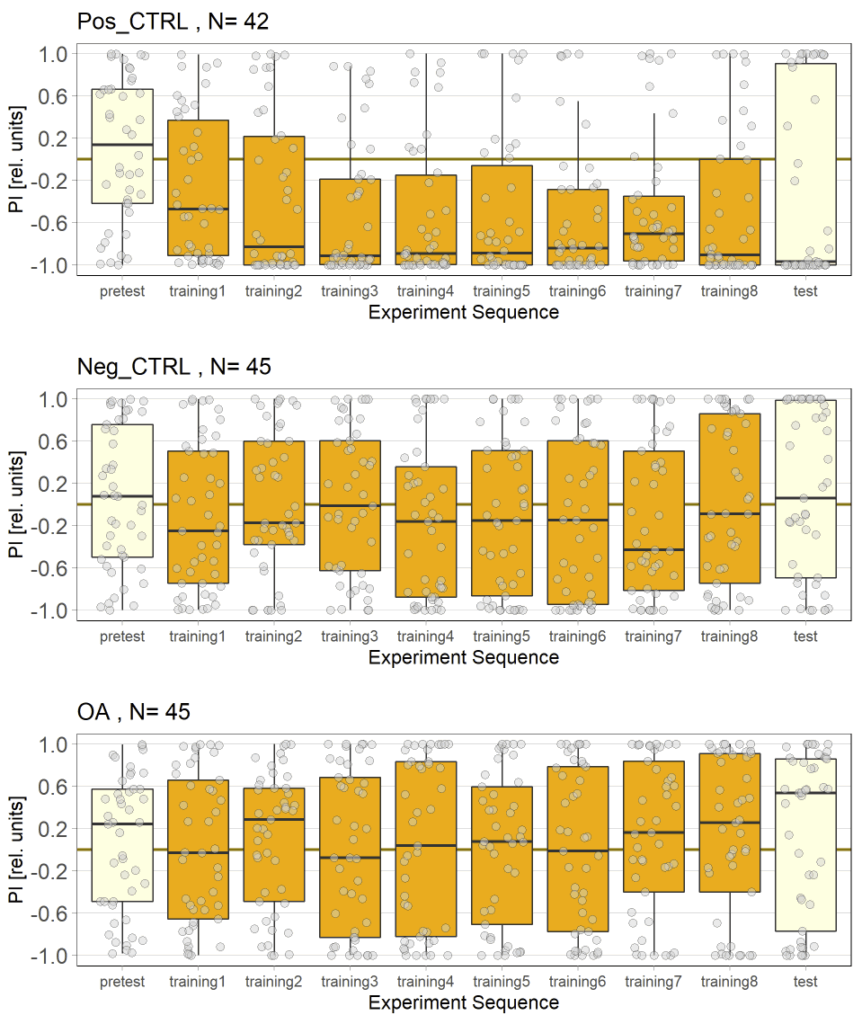
Category: Optogenetics | No Comments
Updated results from JoyStick experiments with the “new” control driver line (and some OA flies)
on Monday, October 13th, 2025 12:46 | by Daniel Döringer
Unfortunately I am still dealing with the problem that flies from my negative control group avoided optogenetic activation, even when their optogenetic channel should not work without ATR supplementation. To tackle this problem I used a “new” effector line (I prepared a new stock from our stock collection) for crossings and tested the offspring, without any improvement concerning the avoidance. Unfortunately the “new” effector line I tested turned out to have lost its “NorpA” mutation which would ensure that the male offspring is blind, thus should rule out any phototaxis-bias. Since flies still avoided the light, a) the effector line does not seem to be the problem and b) the ability to see does not seem to affect the flies behavior in the JoyStick setting.
As a next step I targeted the driver line, as maybe a mutation might have lead to an increase in Gal4. Since we always observed a slightly negative values for our negative control group, hinting that at least some residual activation of the CsChrimson channel is possible even without ATR, an increase in Gal4 might lead to a higher expression of the optogenetic channel.
Residual activation + More channels = More residual activation = Our observed avoidance???
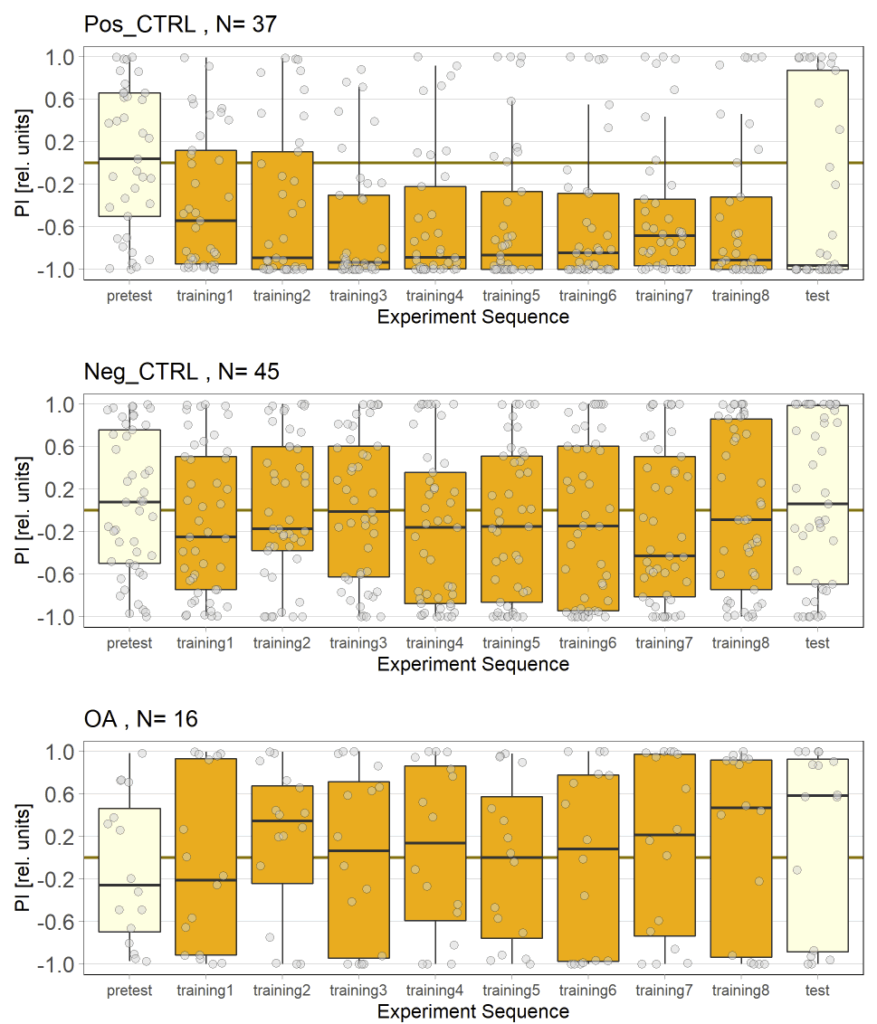
So positive control looks good, negative control is still slightly negative but to an extend that I would consider neglectable.
The additional group here called “OA” are OA;VuMA2 x NorpA, 20xUAS-Chrimson flies. It’s way too early to make any assumptions from the current data but I am looking forward to the results for this group.
Update 27/10/25: Added more flies
Category: Biogenic Amines, Optogenetics | No Comments
Salt (1.5 M) avoidance of 3rd instar larvae
on Sunday, October 12th, 2025 11:53 | by Radostina Lyutova
Category: Biogenic Amines, DAN, Food preference, Larve, neuronal activation, Optogenetics | No Comments
Salt (1.5 M) avoidance of 3rd instar larvae
on Sunday, September 28th, 2025 12:21 | by Radostina Lyutova
Category: Biogenic Amines, DAN, Food preference, Larve, Mushroom Body, neuronal activation, Optogenetics | No Comments