Flexible Valence Coding by Dopaminergic PPM2 Neurons in Drosophila
on Monday, January 26th, 2026 12:11 | by Daniel Döringer
Dopaminergic systems are involved in various physiological processes including motivation and valuation. Studies on Drosophila melanogaster imply that certain insect dopaminergic neurons (DANs) are central for the mediation of valuation, as activity in these neurons can substitute for teaching signals in classical conditioning. This view may oversimplify the complex circuitry of the fly’s dopaminergic system. We focus on an understudied population of DANs and their involvement in valence coding. Using a combination of optogenetics and different operant self-stimulating paradigms, we investigated whether flies expressing an optogenetic channel in DANs of the posterior protocerebral posterior medial cluster (PPM2) would approach or avoid optogenetic stimulation. Flies’ choice was not consistent over the course of our experiments: Initially, animals avoided the stimulating light but this avoidance weakened over time and shifted to mild approach behavior in the final stages. Pharmacologically impairing DA synthesis abolished both effects. Actively exposing flies to the stimulating light aimed to identify whether the valence shift was due to accumulating neural signaling/dopamine release during the experiments. However, exposing flies to light before testing did not induce any preference changes in one-minute choice, hinting that the behavioral change does not occur from prolonged signaling in these neurons. The observation that the very same dopaminergic neurons can mediate both, avoidance and approach behavior in our self stimulating experiments, challenges a central, dopamine-mediated valuation system. Additional findings from flies that express the optogenetic channel in heat-sensing neurons further weaken the claim of centralized dopaminergic punishment neurons, as heat-punishment was independent of dopamine.
Category: Optogenetics | No Comments
Optogenetic activation of Mushroom body Kenyon cells affects naïve salt avoidance in Drosophila larvae
on Monday, January 26th, 2026 11:58 | by Radostina Lyutova
The mushroom bodies (MBs) are known to be the most prominent olfactory memory center in Drosophila. The MB Kenyon cells receive olfactory (via projection neurons, PNs) and gustatory (via dopaminergic neurons, DANs) input. The coincident detection of these stimuli elicits then the formation of associative memory. However, little is known whether and how this high order brain center affects naïve behaviors such as olfaction and gustation. We use fruit fly larvae to test the innate salt avoidance in a simple choice assay. Drosophila larvae are tested for 3 minutes for their preference for 1.5M NaCl. We show that salt avoidance is enhanced upon optogenetic activation of the Kenyon cells. This effect was also observed after ablation of pPAM DANs with simultaneous optogenetic activation of the KCs. We were able to phenocopy this effect by optogenetic activation of TH-Gal4 positive dopaminergic neurons, however not after artificial activation of single DANs of the DL1 cluster. In contrast, larvae showed no salt avoidance after optogenetic activation of sNPF-Gal4 positive neurons. Neuropeptidergic and dopaminergic signaling are tightly involved in modulation of the MBs. However, it is not clear whether our observations depend on coherent neuronal pathways. Therefore, we use simultaneous manipulations of different components of the MB, dopaminergic and sNPF circuitries to unravel the neural mechanisms of modulation of naïve gustatory behaviors in the Drosophila larvae.
Category: Biogenic Amines, DAN, Food preference, Kenyon cells, Larvae, Mushroom Body, neuronal activation, Optogenetics, PAM | No Comments
TH-C-AD; TH-D-DBD x UAS-CD8::GFP Stainings
on Sunday, January 18th, 2026 3:48 | by Daniel Döringer
Category: Anatomy, Optogenetics | No Comments
T-Maze experiments with Yellow light and 5 min of previous exposure
on Monday, January 12th, 2026 12:54 | by Daniel Döringer
Category: Optogenetics | No Comments
First T-Maze trial with previous light exposure
on Monday, December 22nd, 2025 12:56 | by Daniel Döringer
Category: Optogenetics | No Comments
Overview PubMed literature analysis for Query “Drosophila optogenetic dopamine neurons behavior”
on Monday, December 22nd, 2025 12:54 | by Daniel Döringer
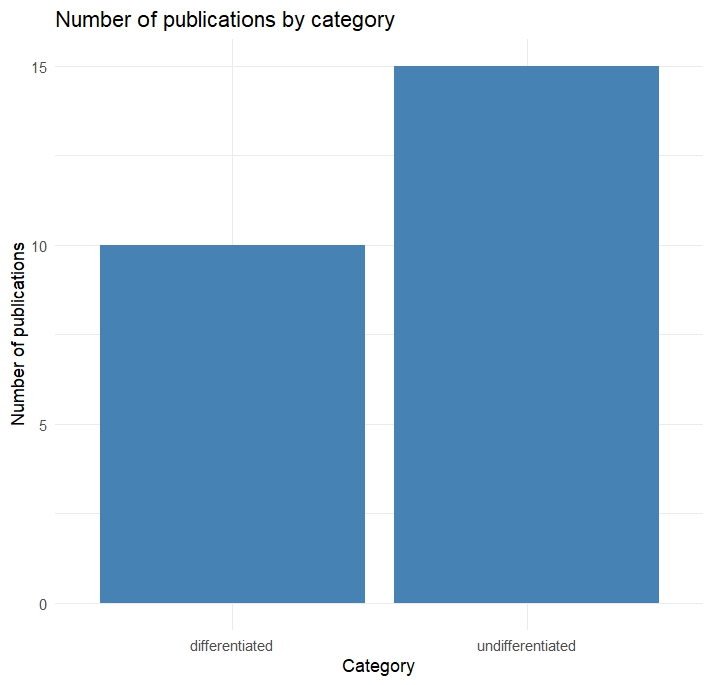
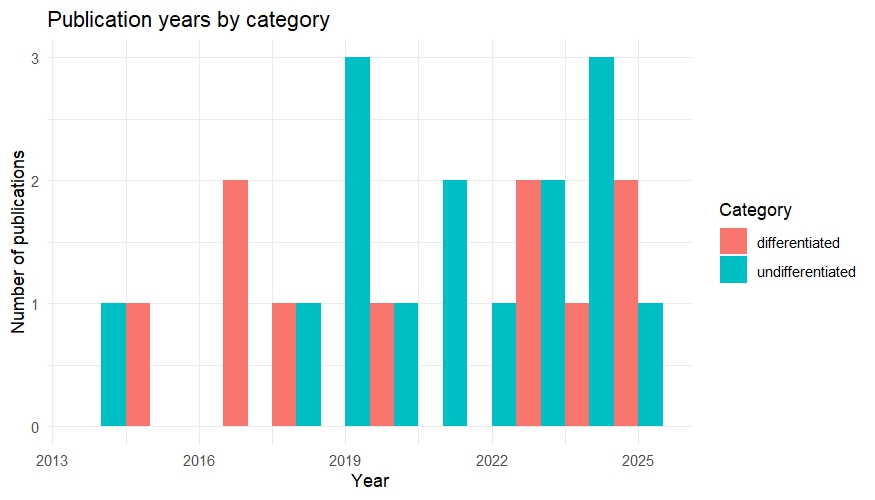
Since this will be a main focus of my master thesis I started a literature analysis to check how the idea of a possible “reward system” in Drosophila is addressed. Based on how authors address this topic, I categorized the results by either “differentiated” or “undifferentiated”. In this case undifferentiated means the authors were addressed certain sets of neurons as “reward neurons” or “punishment neurons”, although they could never be classified in this broad sense. For example a lot of papers of the “undifferentiated” category address dopaminergic neurons from the PAM cluster as “reward” neurons, since flies formed appetitive olfactory associative memories, when the CS (odors) were paired with optogenetic activation these neurons. Since PAM neurons are activated during sugar detection it makes sense that they can substitute for reward, but that doesn’t make them “reward neurons”. Real “reward neurons” would activated in any instance of reward, independent of whether it is sugar, mating or water. Many authors do not address this issue properly. Although this analysis is not finished yet I created two plots to visualize the general trends:
Category: Optogenetics, writing/publishing | No Comments
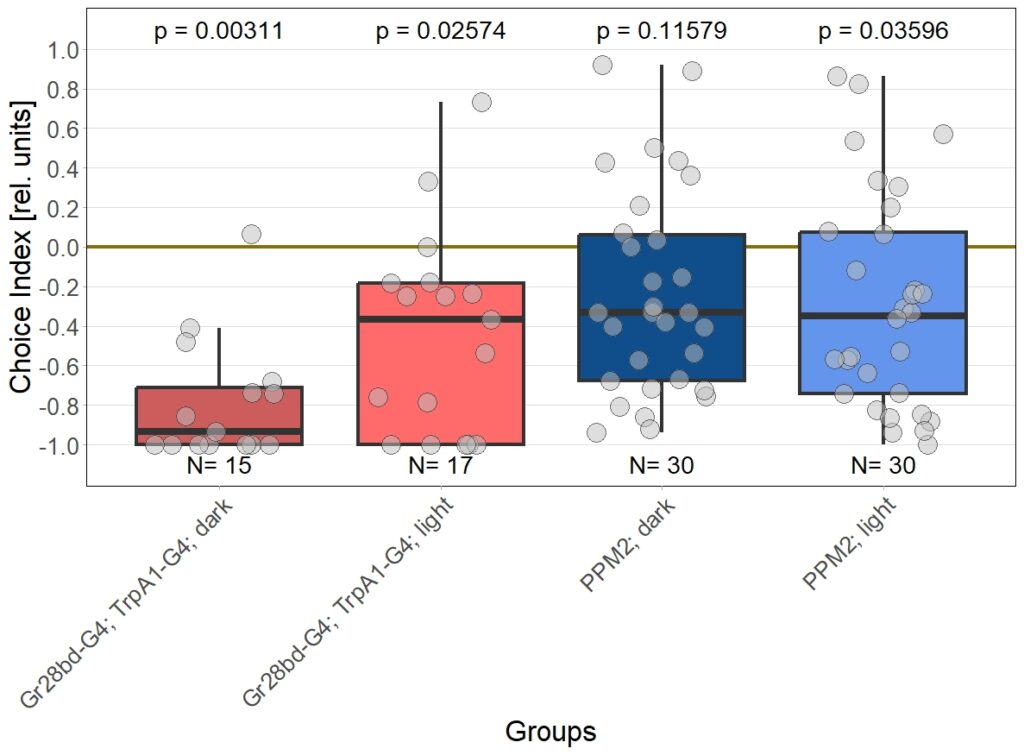
Final results of yellow light T-Maze experiments with dopamine depletion using 3IY
on Saturday, December 13th, 2025 6:15 | by Daniel Döringer
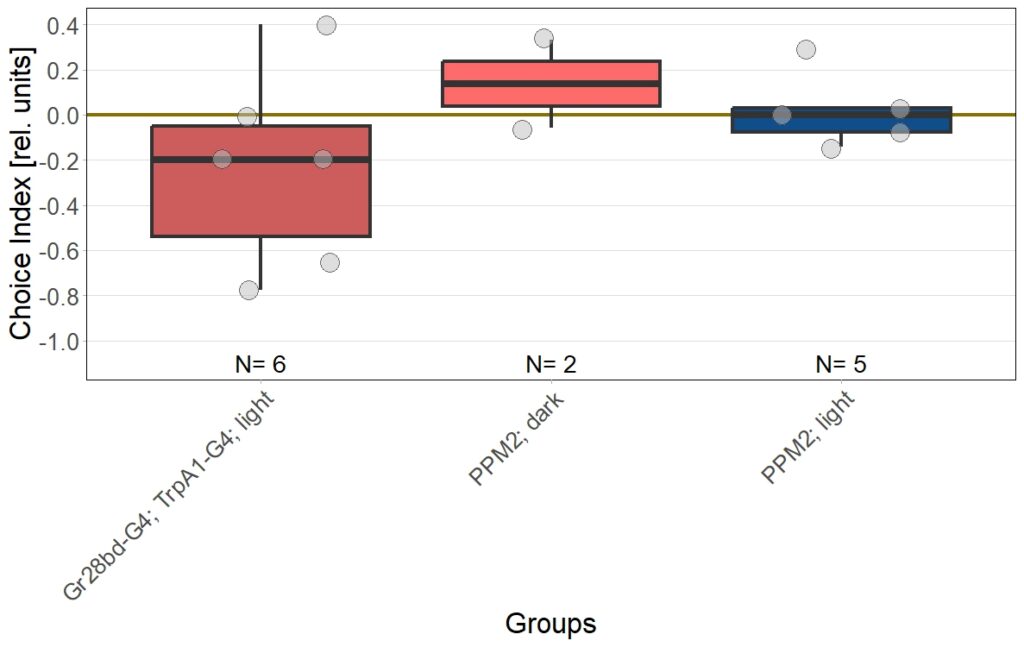
In previous experiments we tested flies expressing the optogenetic CsChrimson channel in PPM2 neurons. We observed mild avoidance at 1 minute, which decreased over the time course of 10 minutes, even leading to positive choice indices. To verify whether the observed effect was instructed by dopaminergic signaling, I depleted flies of dopamine using the competitive tyrosine-hydroxylase inhibtor 3-Iodo-L-Tyrosine, and tested flies again in red and yellow light T-Maze, for 1 and 10 minutes. The figure below shows the results for the yellow light T-Maze. Although we can see the same trend from avoidance in the beginning to mild approach after 10 minutes, this effect was not significant. For the 3IY treated experimental groups, we can assume that the general effect mediated by these neurons seems to be absent, when animals are depleted of dopamine.
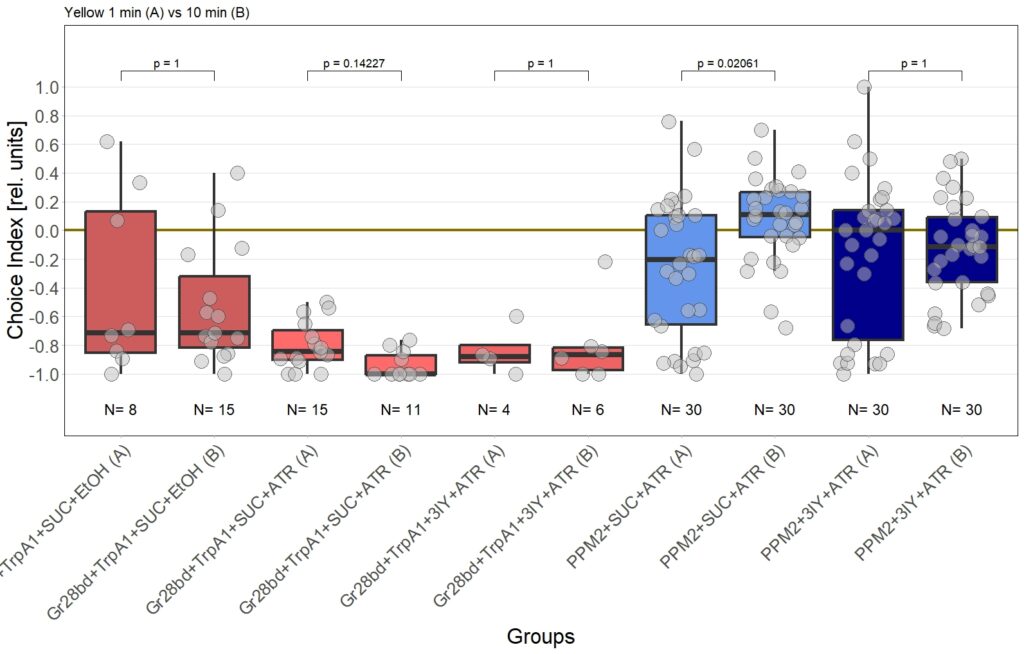
Although I could not observe a significant effect here, I plan to follow up these experiments with yet another set of T-Maze experiments. This time I plan to actively activate the neurons prior to testing. I hypothesize, that prolonged activation of the neurons might affect their valence for the animal and therefore expect animals which experienced this activation to show differences in 1 minute testing.
Category: Optogenetics | No Comments
Current T-Maze results and power analysis
on Friday, December 5th, 2025 2:35 | by Daniel Döringer
I am currently still performing T-Maze experiment with red light, to check for dopamine dependency of the effect I observed in the original 1 vs. 10 minute T-Maze experiments using yellow light and PPM2 flies.
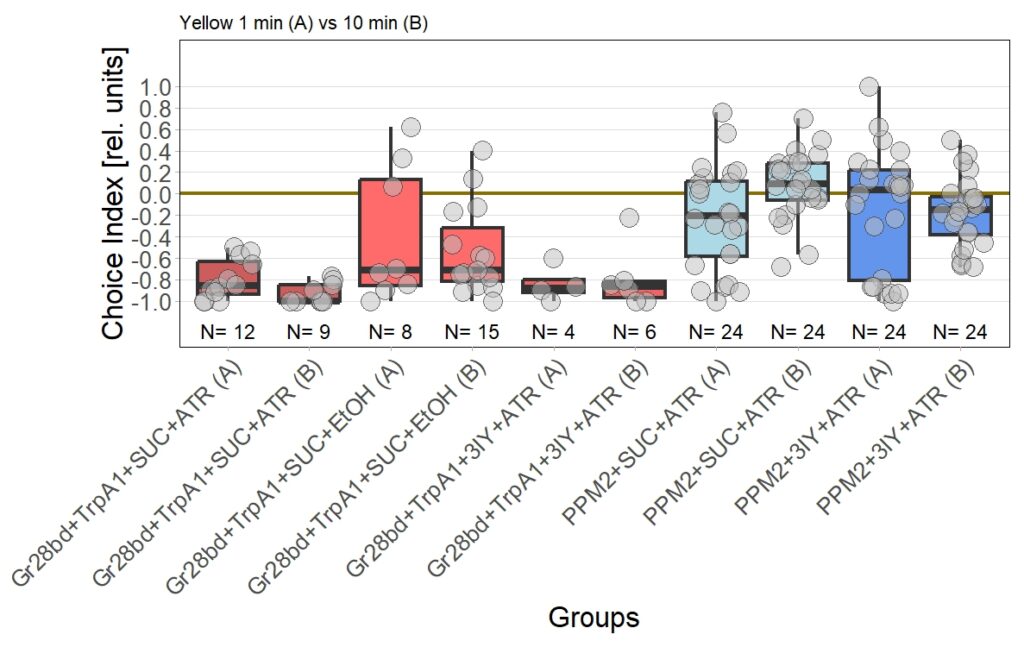
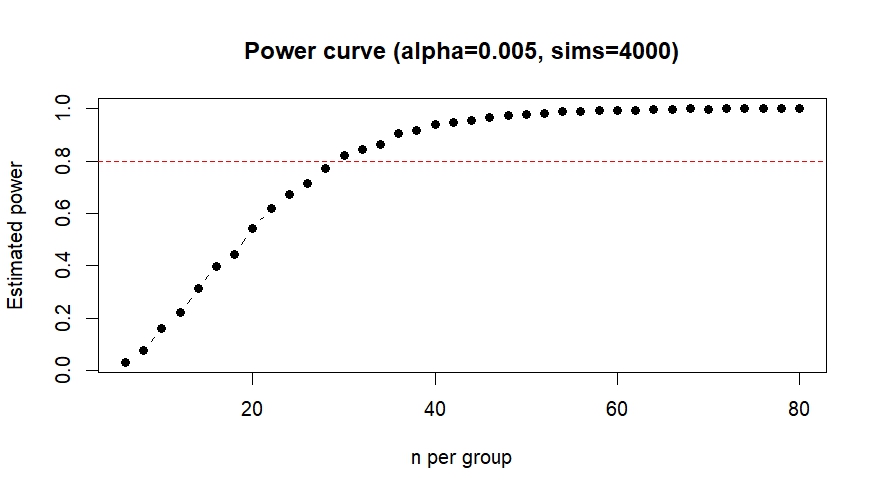
For now I planned to aim for a sample size of 30 experiments per experimental group, with the justification that this was also was I aimed for in the original screen. To see whether this sample size is sufficient to verify the effect (or it’s absence) I performed a power analysis and based it on the previously observed effect size.
Cohen’s d (from original screen) = -1.023 (large effect)
I used the power.np.wilcoxon() function from the pwrss package to calculate the sample size needed to achieve 80% statistical power with an alpha level of 0.005.
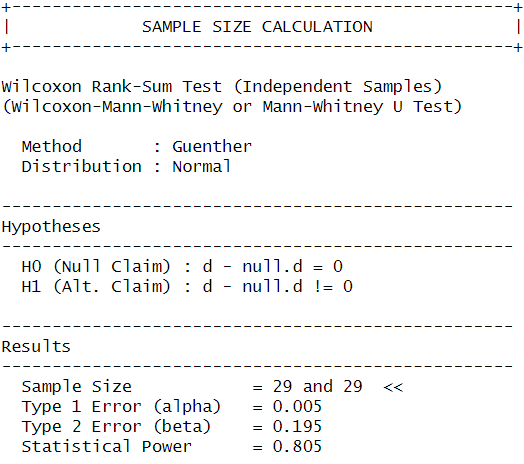
This confirmed that I would need at least 29 observations per group to detect the same effect as observed before, so I will keep aiming for a sample size of 30. Since I was unsure whether my analysis was correct I performed a second power analysis in R with an analysis script provided by ChatGPT, which calculated the sample size using simulated drawings from my original data. This analysis also resulted in a target sample size of 30 so I am fairly confident my calculations were correct.
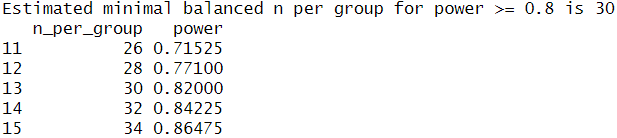
Category: Optogenetics | No Comments
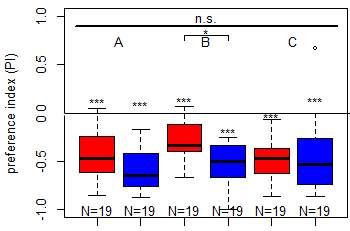
Salt (1.5 M) avoidance test in TH-D1-Gal4 larvae under red and blue light [N = 19]
on Sunday, November 23rd, 2025 2:43 | by Christoph Kumpfmüller
Category: crosses, Larve, Mushroom Body, Optogenetics | No Comments
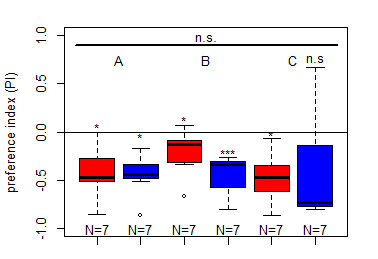
Salt (1.5 M) avoidance test in TH-D1-Gal4 larvae under red and blue light
on Saturday, November 15th, 2025 2:53 | by Christoph Kumpfmüller
Category: DAN, Larve, MBON, Mushroom Body, Optogenetics | No Comments