Tdc2 and Bruchpilot in flight
Tested the third cross. Now, it is around 15n each group. After a control experiment I preferred a recovery time of 1h instead of 5h. A short pilot for control groups showed that Tdc2virgins crossed to w1118males have a flight defect.
3rd
“Virtual brain” site
Arnim Jenett (Janelia Farm Research Campus), Kazunori Shinomiya, Kei Ito (both Tokyo University), and other anatomists made a great site with a 3D-viewer of adult Drosophila brains available. You have the chance to scroll threw a whole mount stack while ticking different brain areas. Those brain areas are listed next to the stack. Different areas are coloured differently, so that you can look at the location of several areas in the same brain. On the main page you can find simply explained tutorials about the usage of the site. It is correlated to the anatomical search engine of the Janelia farm GAL4 collection.
Because it was very helpful to me to learn all the synonyms of relevant areas and because I think it is very helpful to learn more about the structure of the Drosophila brain in general I wanted to advertise the site here.
https://www.virtualflybrain.org/site/vfb_site/overview.htm
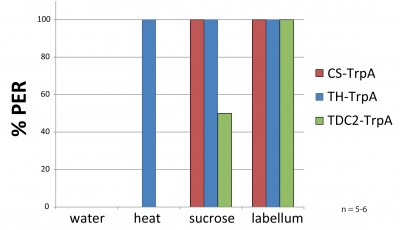
Usage of TrpA with infrared lamp
As a pilot for future experiments I tried to reproduce results from Keene et al., 2012. There, they substituted sugar by a laser to provoke PER after 24h of starvation (females, one week old). In TH-GAL4 they found 100% response when they pointed the laser to head or thorax. In tdc2-GAL4 indeed they found 50% of flies responding to heat but only when the laser was pointed to the thorax but not to the head.
Since we do not have this kind of laser, they used, I played around with an infrared light. Flies were fixed to hooks and a clamp as always. First, there was given a filter paper soaked with EVIAN-water (negative control), then there were 10s of heat (36-38°C), afterwards two positive controls: 30% sucrose on the legs and finally on the labellum.
In the figure, you can see that I am able to reproduce the methods with an infrared lamp instead of a laser. The TH-GAL4 flies crossed to TrpA respond much more to heat compared to the controls (see figure in blue and red). The proboscis extension appears already after a few seconds.
Unfortunately, I could not reproduce the results for tdc2-GAL4 (see figure in green). There was no response to heat found at all. (tdc2-GAL4 has to be ok, because I see fluorescence when crossed to GCaMP). Interestingly, the flies did not respond as strong to the 30% sucrose as expected. The starvation may have to be longer to increase motivation?
Next step will be to see a phenotype that can be seen by activating tdc2-GAL4. That may be PER after longer starvation or flight behavior.
Heat shock rescue of tßh-gene in Buridan’s paradigm
The loss of tßh in adult flies leads to decreased walking speed and an increase in stripe fixation. Rescuing the gene by a heat shock construct (flies from Henrike Scholz, Cologne) increases walking speed back to wild type level but cannot change stripe fixation.
It is possible that the phenotype in stripe fixation is not exclusively tßh-dependent but more due to other problems the tßh-mutants have, e.g. developmental defects. That would ask for another heat shock timing.
Results:
– 1st cross
– 2nd cross
Flies:
– 1st cross (with heterozygous controls):
males females females
w1118,tßh/y ;; HStßh x w+,tßh//FM7 -> w1118,tßh//w+,tßh ;; HStßh//+
w1118,tßh/y x w+,tßh//FM7 -> w1118,tßh//w+,tßh
w1118,tßh/y ;; HStßh x w+ -> w1118,tßh//w+ ;; HStßh//+
w1118,tßh/y x w+ -> w1118,tßh//w+
– 2st cross (only mutants):
w1118,tßh/y ;; HStßh x w+,tßh//FM7 -> w1118,tßh//w+,tßh ;; HStßh//+
w1118,tßh/y x w+,tßh//FM7 -> w1118,tßh//w+,tßh
Heat shock timing:
Trehalose measurement in Würzburg with Dennis Pauls
Draft of protocol
Material:
0.5 µl capillary with adequate pump, 0.5ml and 1.5ml Eppis, ice, peaked stylus, razor blade, 2 forceps (or scissors)
Procedure:
sting 3 holes in a small eppi (size is important, not to small so that enough hemolymph can go through, not to big so that no fat or other dirt is going through), put into big eppi.
20 adult flies on ice, cut their wings (not sure about the reason)
spear the fly’s thorax with the peaked stylus
store flies in the small eppi with holes
centrifuge the small eppi within the big one (1 min, 5000 rpm)
throw away small eppi, cut big eppi (to access the pellet easier), soak the pellet with a capillary
record the amount of soaked hemolymph with a ruler (e.g. by photographing under disscetion scope)
pump the hemolymph out of the capillary into a fresh small eppi and freeze it.
Remarks:
Everything has to be done on ice, cooling chain should as poosible not be interrupted
(5000 rpm can be differ with different centrifuges…)
First flight tests, Trehalose again…
Chrisi’s day…
1 Collected virgins for our ill post-doc, made some crosses:
d42-GAL4 males x tubGAL80-UAS-PKCi and x CS
d42-chaGAL80 males x tubGAL80-UAS-PKCi and x CS
2 Buridan: Prepared colored food (10mg/ml, 2.5ml total in the very little vials, ~ 5 flies in each vial). Denise tested in the afternoon females in the Buridan. Data (4n now) will be shown in the lab meeting.
3 Imaging Made a new cross for the Imaging-project:
TDC2-GAL4 males x UAS-GCaMP3(2)
replaced Neloy’s flies by flies from stock
Florian’s larval preparation (good!) showed that GCaMP-expression is much less than GFP-expression. They hope for the homozygous line.
4 Flight: Performed flight experiments with the tbh,TDC,UAStbh flies. Raw data, see below. Thinking about analysis (probably tomorrow)
Protocol: glued the day before, sugar in wheel chambers, little EVIAN-water.
5s filter on the legs before starting, aspirated, stopped when no response 3 times in a row, stopped time when 30min, stopped after 15 aspiration trials
| date | fly | genotype | number_aspiration | time_to_first_stop | duration_of_flights | remarks |
| 01.11.2012 | 1 | 12 | 4.56 | 4.56,0.22,0.30,0.2 | ||
| 01.11.2012 | 2 | 2 | 49.15 | 49.15,14.42 | ||
| 01.11.2012 | 3 | 16 | 0.03 | 0.03,0.15,3.06,1.56,0.3,0.14,4.58,0.22 | ||
| 01.11.2012 | 4 | 3 | 0 | 0 | ||
| 01.11.2012 | 5 | 40 | 0.05 | 0.05,0.01,0.02,0.01,0.01,0.02,0.02,0.01,0.02,0.03,0.02,0.02,0.01,0.01,0.01,0.01,0.01,0.04,0.09,0.01,0.02,0.01,0.11,0.01,0.01,0.01,0.01,0.01,0.02,0.01,0.03 | ||
| 01.11.2012 | 6 | 3 | 0 | |||
| 01.11.2012 | 7 | 13 | 3.01 | 0.01,0.01,0.01,0.01,0.01,0.02,0.01 | ||
| 01.11.2012 | 8 | 15 | 0.01 | 0.01,0.04,0.03,0.02,1.56,0.15,0.02,1.46,3.26,1.15,0.46,6.45,2.56,0.06,12.59 | ||
| 01.11.2012 | 9 | 15 | 0.01 | 0.01,0.01,0.28,0.1,1.5,1.29,0.40,0.29,1.34,0.54,0.22,0.37,0.31,0.54 | ||
| 01.11.2012 | 10 | 9 | 0.01 | 0.01,0.01,0.01,0.01,0.01 | ||
| 01.11.2012 | 11 | 12 | 0.01 | 0.01,0.01,0.01,0.01,0.01,0.01 | ||
| 01.11.2012 | 12 | 3 | 0 | |||
| 01.11.2012 | 13 | 4 | 0.06 | 0.06,0.5,0.06,38.2 | ||
| 01.11.2012 | 14 | 3 | 0.19 | 0.19,0.33,0.31,30 | ||
| 01.11.2012 | 15 | 15 | 3.38 | 3.38,2.43,0.47,0.36,4.06,0.39,1.25,0.35,0.52,1.07,0.22,0.23,0.27 | ||
| 01.11.2012 | 16 | 3 | 0 | |||
| 01.11.2012 | 17 | 3 | 7.16 | 7.16,23.27,30 | ||
| 01.11.2012 | 18 | 3 | 0 | |||
| 01.11.2012 | 19 | 6 | 1.33 | 1.33,14.57,9.17,30 | ||
| 01.11.2012 | 20 | 1 | 18.20 | stopped the expriment | ||
| 01.11.2012 | 21 | 3 | 2.50 | 2.50,2.18 | stopped the expriment |
5 Trehalose: Found time to try out Gérard’s idea (no pigment in the probes to not disturb the NanoDrop measurement). 4 groups: 10 flies with or without heads, 50 flies with or without heads. Hilde showed me how to remove heads with liquid nitrogen and vortex. The other flies (with heads) were frozen at -20° as usual. Trehalase incubation over night. Tomorrow glucose oxidase and measurement
First lab book post
Hi all, I forgot about the lab book email, because it reached me in New Orleans… My day:
1 Collected virgins for our ill post-doc
2 Prepared colored food (10mg/ml, 2.5ml total in the very little vials, ~ 7 flies in each vial). Vicky tested in the afternoon females in the Buridan. The the duration of drug application was increased to 3h because there was no effect after 1 hour. Data will be shown in the lab meeting.
3 Answered to Dennis Pauls to make an appointment for the Trehalose measurement in Würzburg
4 Glued hooks for a flight experiment I did not do because I did not know, how… (idea was to test whether the UAS-tbh cross is ok)
5 Checked my TDCxGCaMP flies for expression in the fluorescence dissection scope (very cool!) in Marco’s room (borrowed from Konstantin). Then realized that there are pupa in the virgin vials :(
6 Temperature-dependent experiments in Buridan: wanted to test the infrared lamp with the dimmer (measure the temperature change). Was impossible because the thermometer itself heated up. So, I finally looked for an infrared thermometer (you can point on the area you want to measure).
7 boring stuff… Dienstreiseabrechnung was wrong… Email to the Chinese guys again about the cylinder for Buridan… Explained Florian (Marco’s master student) some fly stuff…
Octopamine less flies survive longer
Octopamine is a biogenic amine involved in insect physiology and behavioral control. In Drosophila, it was suggested to be necessary for appetitive olfactory learning (Schwaerzel et al., 2003). The available fly mutant tßh cannot convert tyramine into octopamine. We have found that the preference of these mutant flies for sugar (tested in a T-maze) as well as the responsiveness to a serial dilution of sugar (tested in a proboscis extension assay) is decreased compared to their genetic control “w+” (unpublished). We wondered whether the flies have different physiological state and hence different motivation, or if octopamine is involved in the neuronal coding of motivation per se. We therefore want to examine the physiological state of these flies.
In a first approach, we starved flies to death in bottles containing only a cotton pad moisturized with water. Dead flies were counted periodically and kept in the bottles. The survival curve shows that the tßh mutant flies die later (Fig.1a): The time point for 50 % of the flies to die is significantly higher in tßh mutant flies than the one of the control w+. Gender had no effect (ANOVA, gender: p=0.13, genotype: p<0.04). Data from male and female were thus pooled in the figures. These results show that starvation affects differently control and mutant flies. Obviously tßh mutants are more resistant to starvation.
The different physiological state could be due to different activity levels and consequently less energy requirements in tßh flies. To test for this hypothesis, we will use a second approach and measure trehalose content in the hemolymph of the flies. It has been shown that trehalose level decreases with starvation (Meunier et al., 2007). Furthermore there is a correlation found between trehalose level and survival (Isabel et al., 2004). A different metabolic rate in tßh mutants may explain a different trehalose content and the higher survival rate after the same starvation time.
In the future, we hope to be able to titrate the physiological state of the fly using the measure of the trehalose content in their hemolymph, in order to test the sugar responsiveness of mutant and control flies with adapted physiological state. This will allow us to separate the role of octopamine on the fly physiology and its role on the fly motivation.