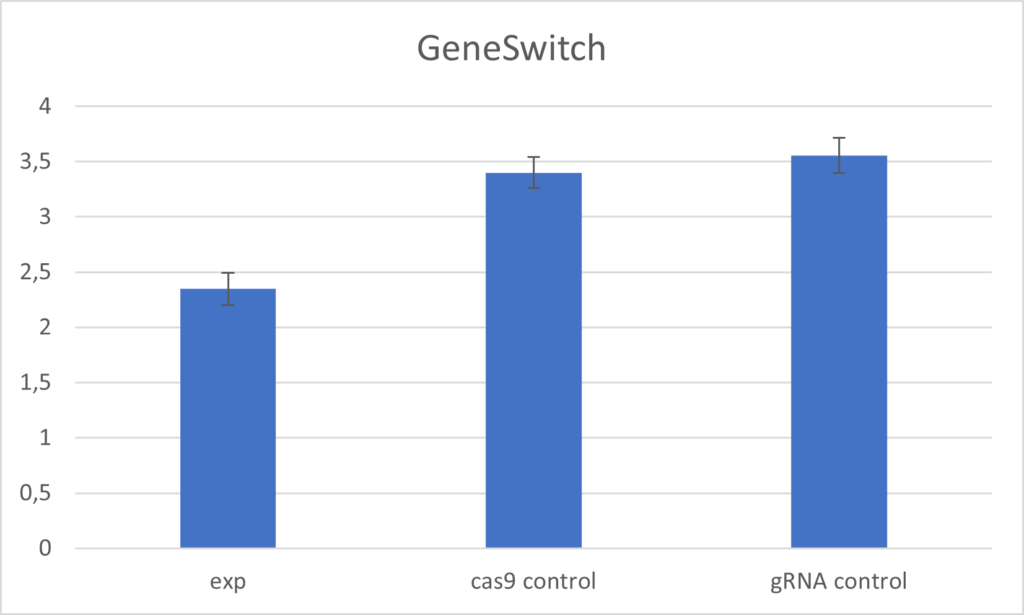
GeneSwitch experiment
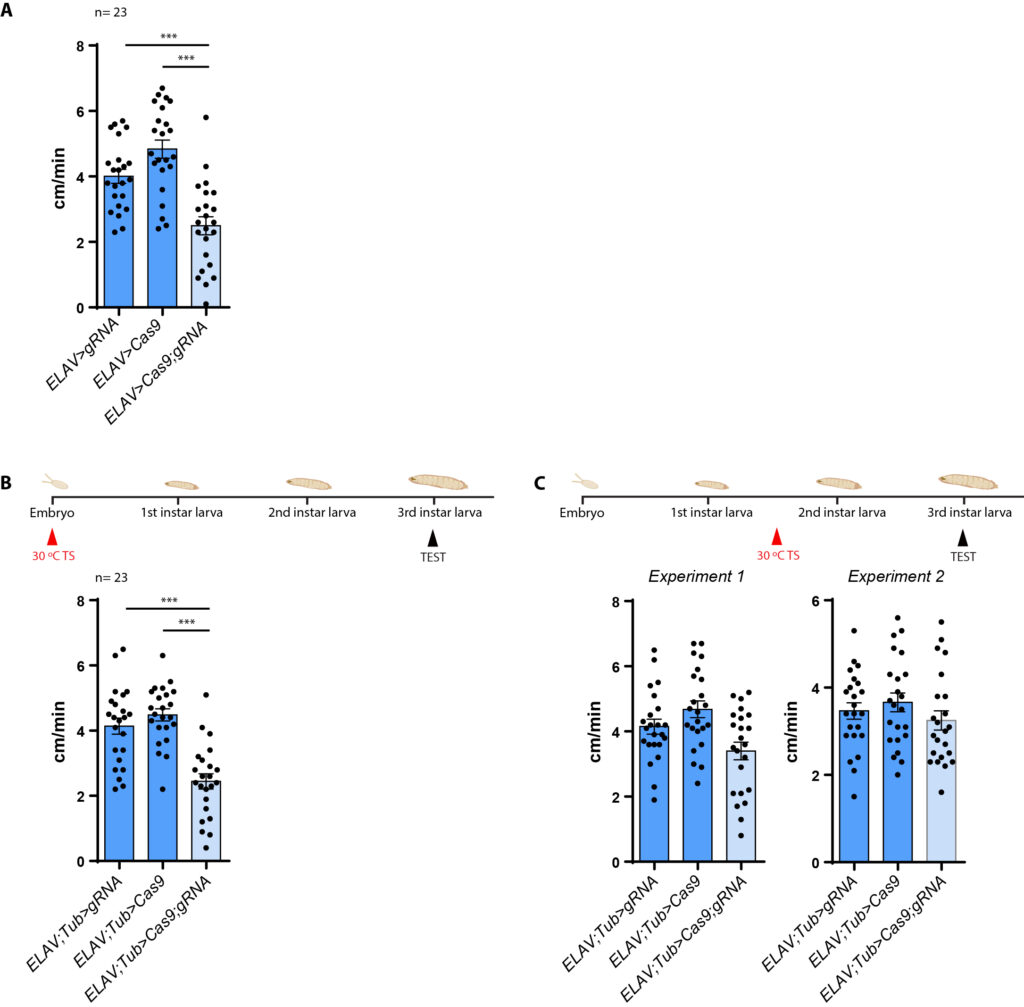
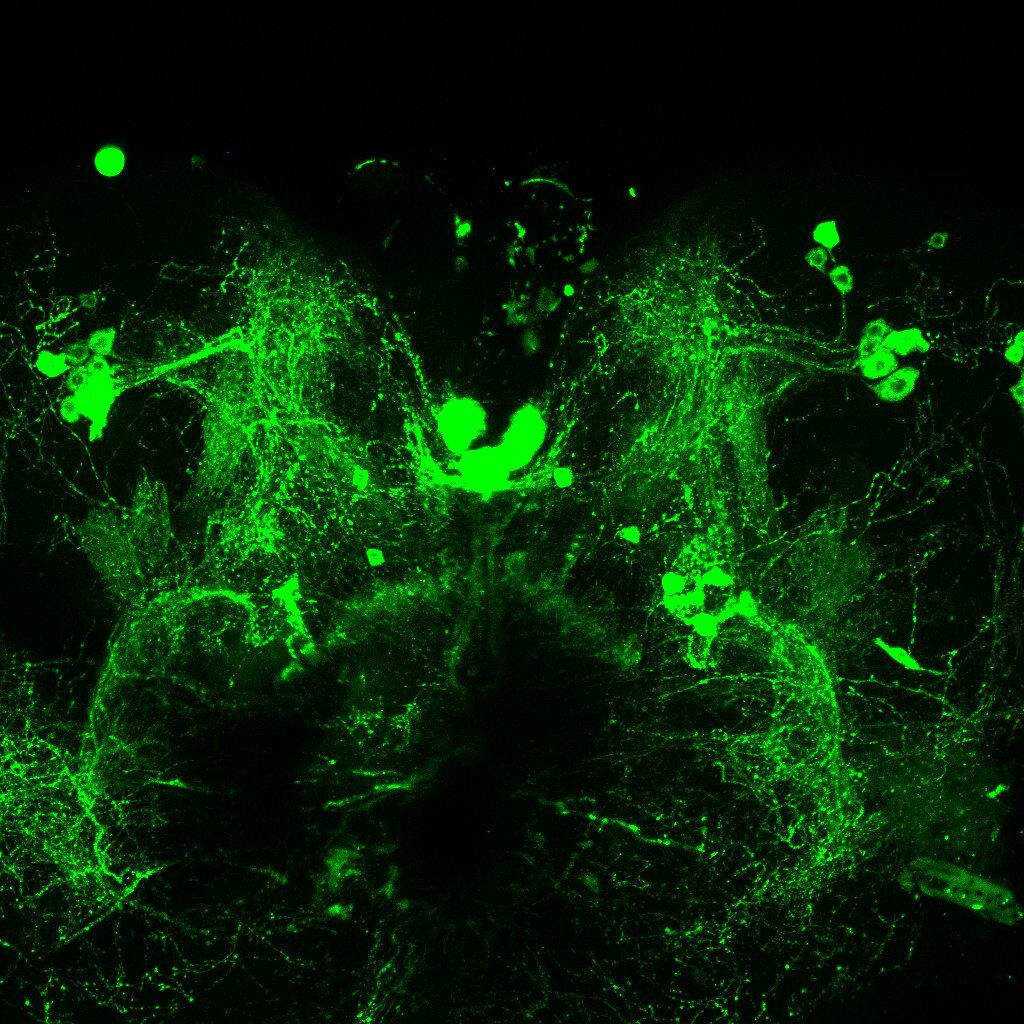
dFoxP knockout in embryonic stage
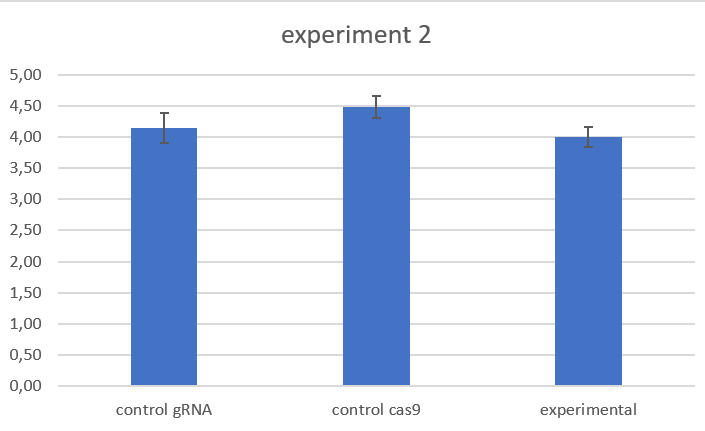
ELAV-Gal4; TubGal80 results
A,B: KO in embyro stage
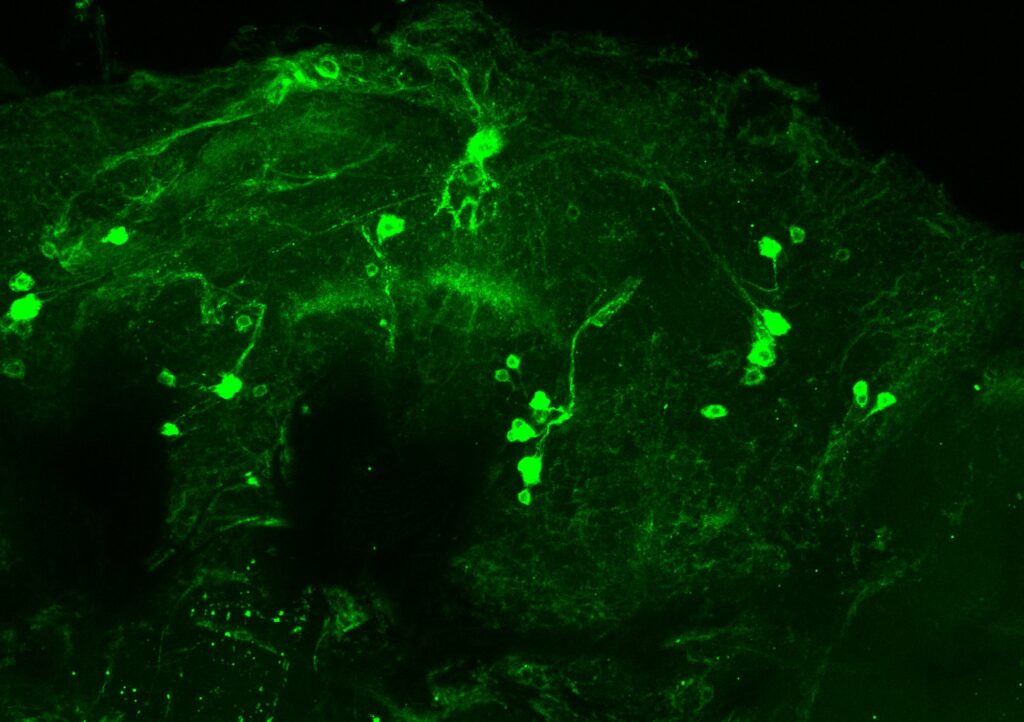
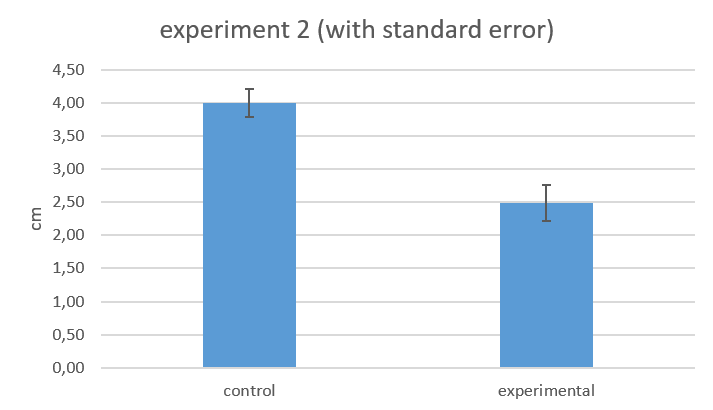
C: KO in larvae stage (Experiment 1: in 30 °C for 18 hours, Experiment 2: in 30 °C for 36 hours)
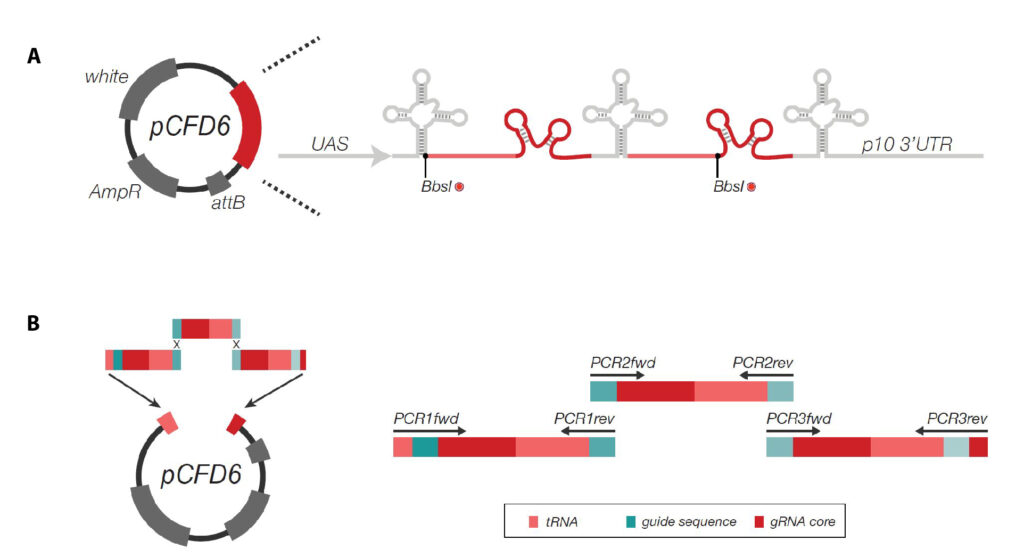
summary of the results of locomotion experiments on drosophila larvae so far and pcdf6 cloning scheme
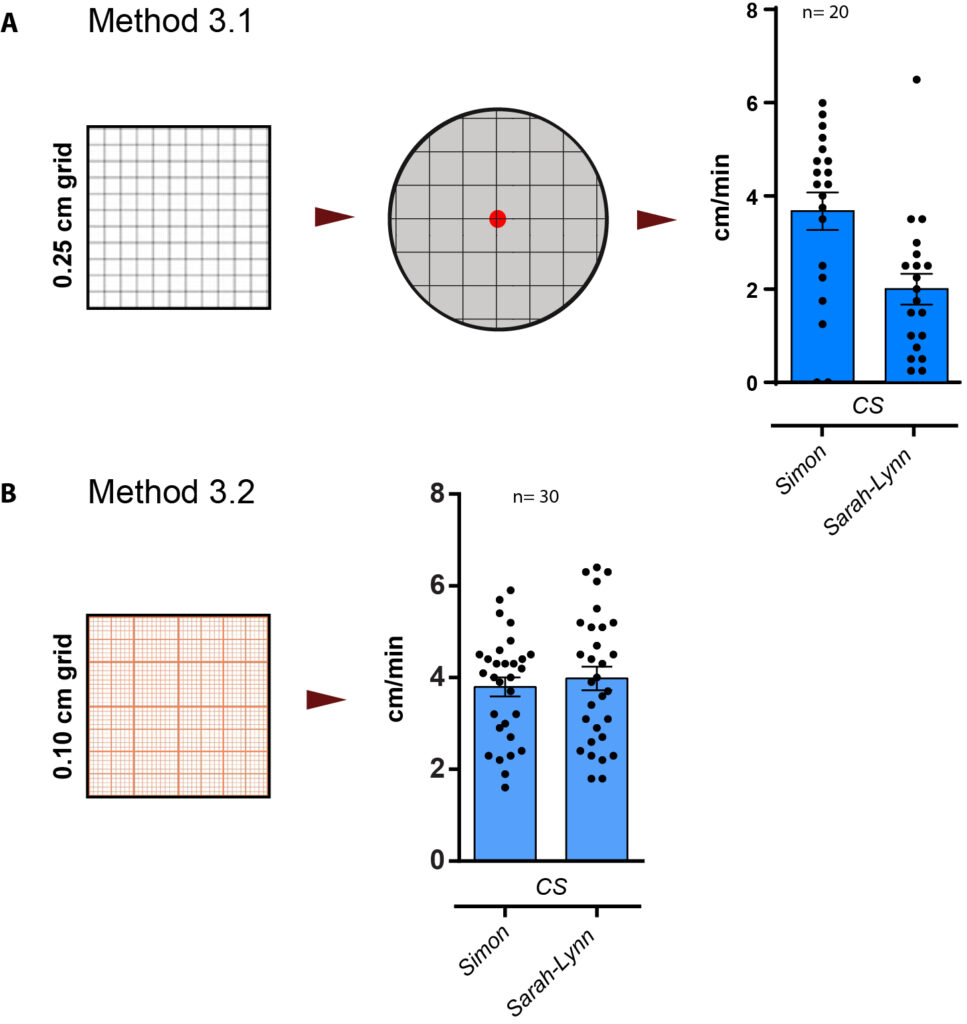
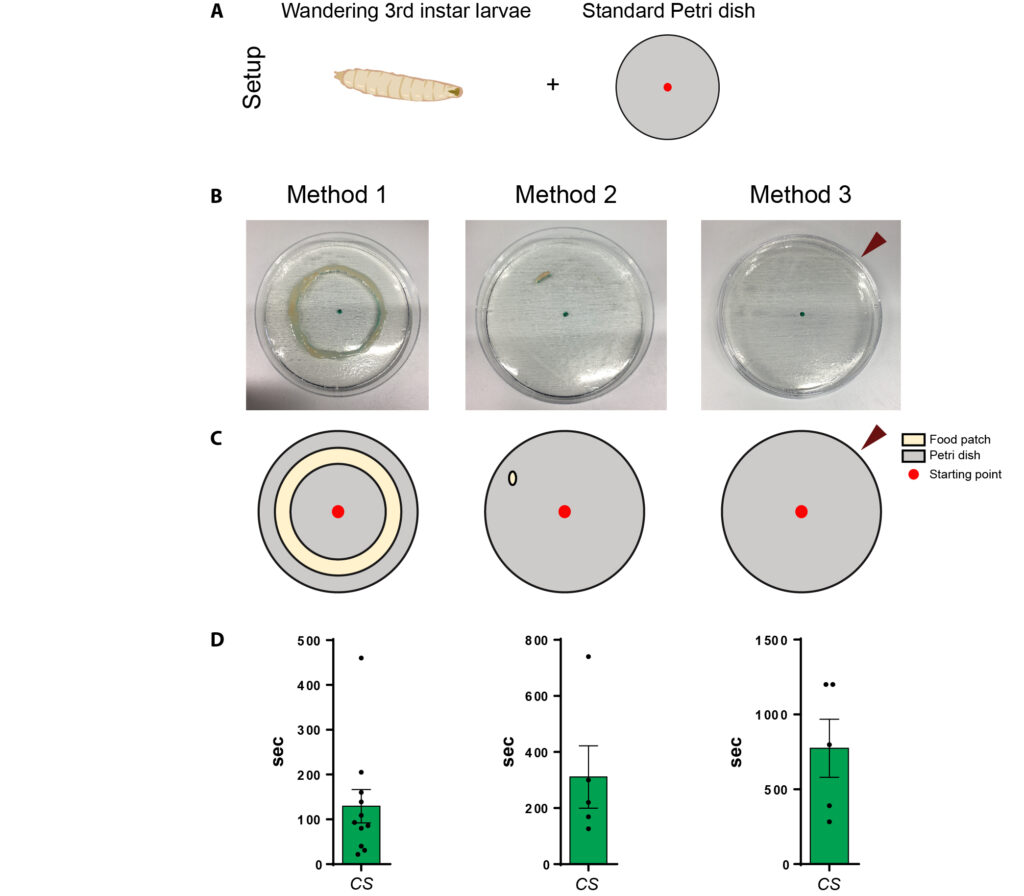
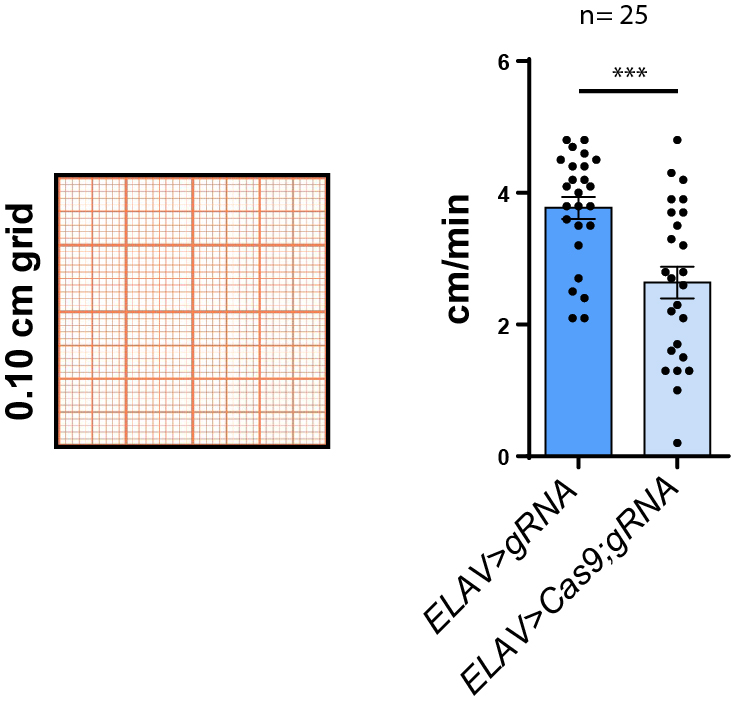
method 3.1: counting squares
0.1 cm grid under microscope, t= 1min
method 3.2: counting sqaures
| Crosses |
| ELAV-Gal4;TubGal80 x UAS-gRNA |
| ELAV-Gal4;TubGal80 x UAS-Cas9 |
| ELAV-Gal4;TubGal80 x UAS-Cas9- gRNA |
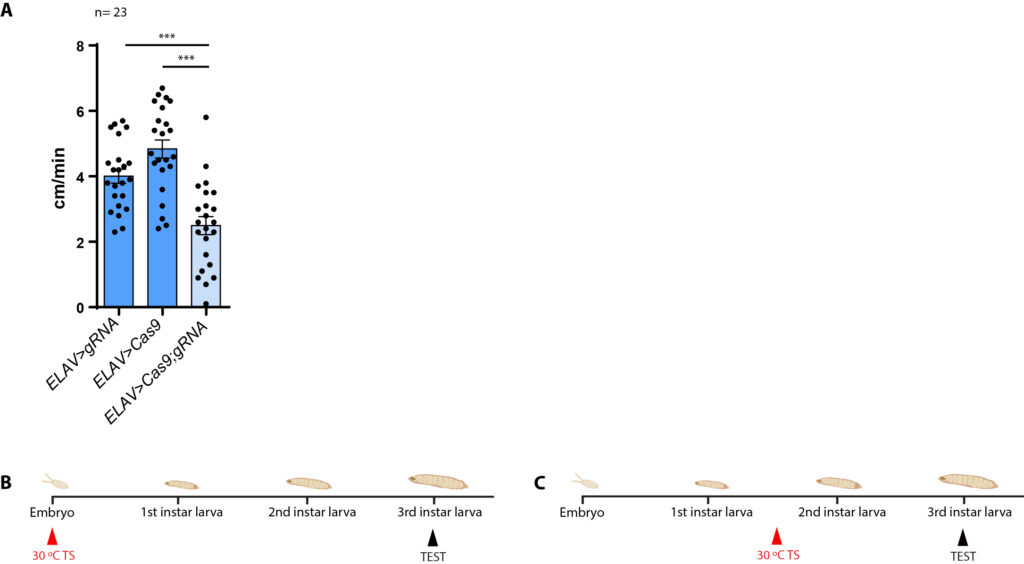
B: larvae-temperature scheme
| Crosses |
| ELAV-geneswitch x UAS-gRNA |
| ELAV-geneswitch x UAS-Cas9 |
| ELAV-geneswitch x UAS-Cas9- gRNA |
Crosses for conditional knockout
| Cross | Temp. | |
| ELAV-Gal4;TubGal80 x UAS-gRNA-Cas9 | 18 °C (day 1-x) | X |
| ELAV-Gal4;TubGal80 x UAS-gRNA | 18 °C (day 1-x) | X |
| ELAV-Gal4;TubGal80 x UAS- Cas9 | 18 °C (day 1-x) | X |
| ELAV-Gal4;TubGal80 x UAS-gRNA-Cas9 | 30 °C | |
| ELAV-Gal4;TubGal80 x UAS-gRNA | 30 °C (day 1-x) | X |
| ELAV-Gal4;TubGal80 x UAS- Cas9 | 30 °C |
further work on pcdf6 cloning, second experiment on larvae locomotion
PCR pCDF6 Primer
-12.5 µl primer reverse/forward
-5 µl dNTPs
-2.5 µl Polymerase
-0.57 µl pCDF6 undigested
-50 µl Q5 buffer
-166.93 µl H2O
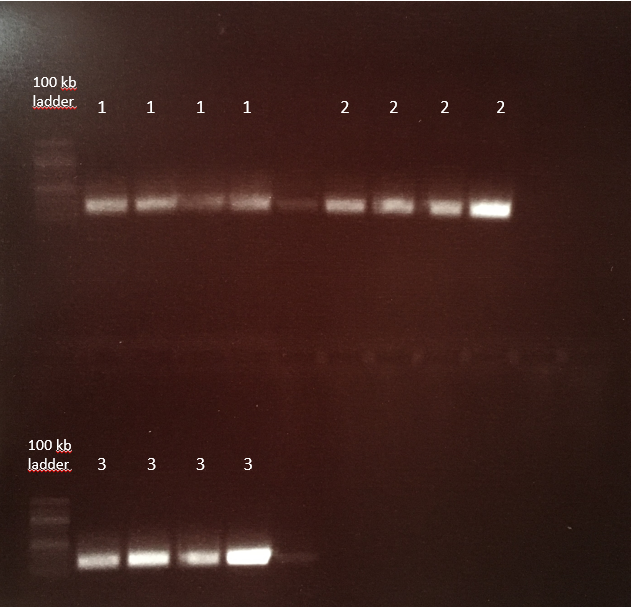
Agarose-gel electrophoresis
E.Z.N.A. Gel Extraction
pCDF6 NEBuilder Assembly Reaction
-pcr1: 4.85 ng -> 0.88 µl
-pcr2: 4.24 ng -> 1.5 µl
-pcr3: 4.87 ng -> 1.6 µl
-pCDF6 digested: 100 ng -> 2.8 µl
-H2O: 3.22 µl
-10 µl NEBuilder HiFi DNA Assembly Master Mix/Control
PCR
heat shock transformation of the construct into competent E.coli cells
-> plated on LB0+Amp plates
-> no colonies
experimental: ELAV-Gal4 x UAS-Cas9-gRNA (n=23)
PCR with undigested pCDF6
primer: reverse/forward each 10 µl
dNTPs: 4 µl
buffer: 40 µl
polymerase: 2 µl
H2O: 133.6 µl
undigested vector: 0.4 µl
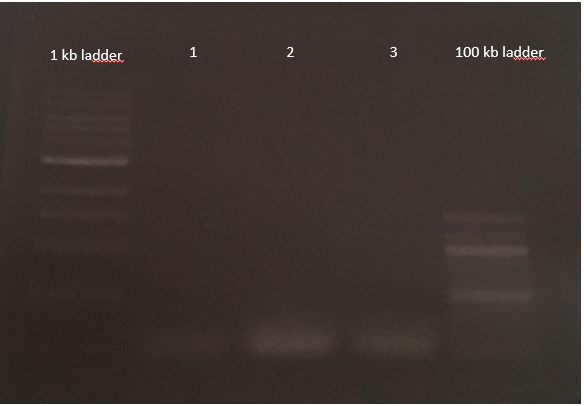
pCDF6 cloning with three inserts
pCDF6 digestion with enzyme Bbs1
-5.5µl vector (undigested), conc.: 539.1 ng/µl
-1 µl enzyme (Bbs1)
-5 µl buffer (CutSmart)
-38.5 µl H2O
Agarose-gel electrophoresis
E.Z.N.A. Gel Extraction-DNA Purification from Agarose gel
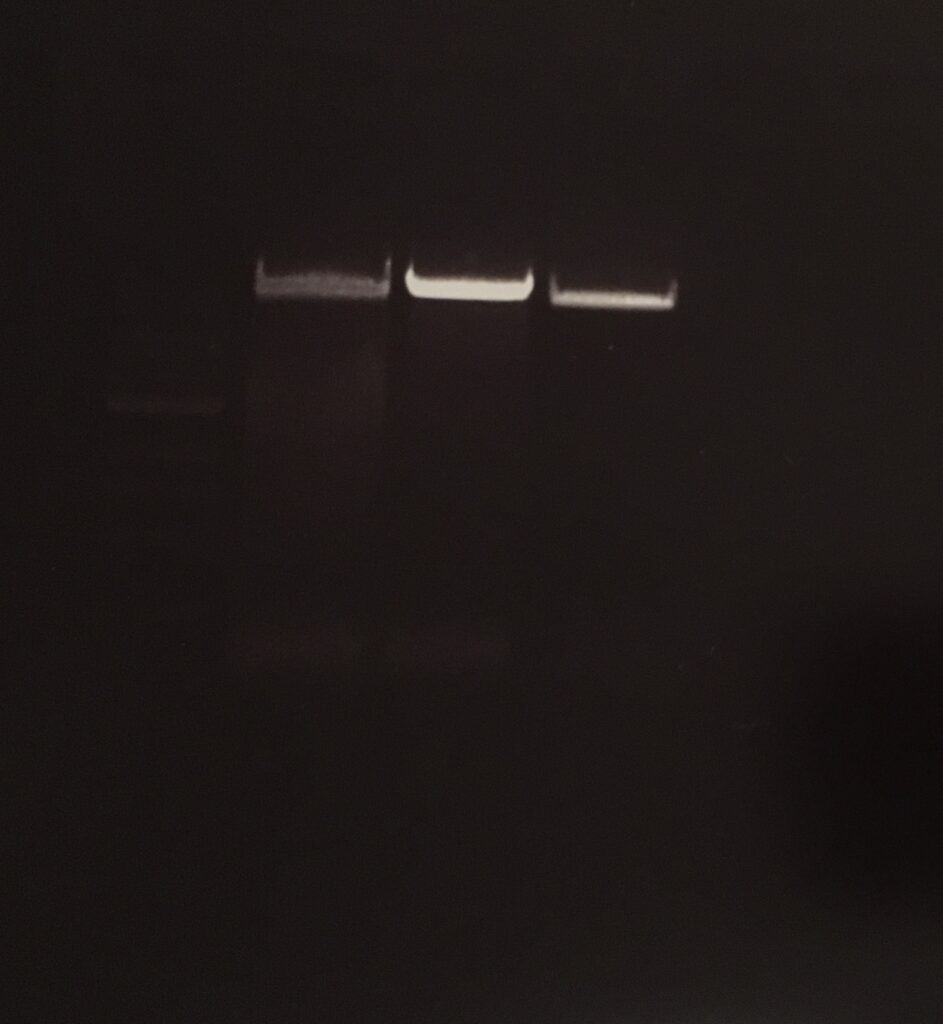
concentration: 38,0 ng/µl
PCR pCDF6 Primer
-12.5 µl primer reverse/forward
-5 µl dNTPs
-2.5 µl Taq-Polymerase
-0.29 µl pCDF6 digested
-50 µl buffer
-167.5 µl H2O
Agarose-gel electrophoresis
E.Z.N.A. Gel Extraction
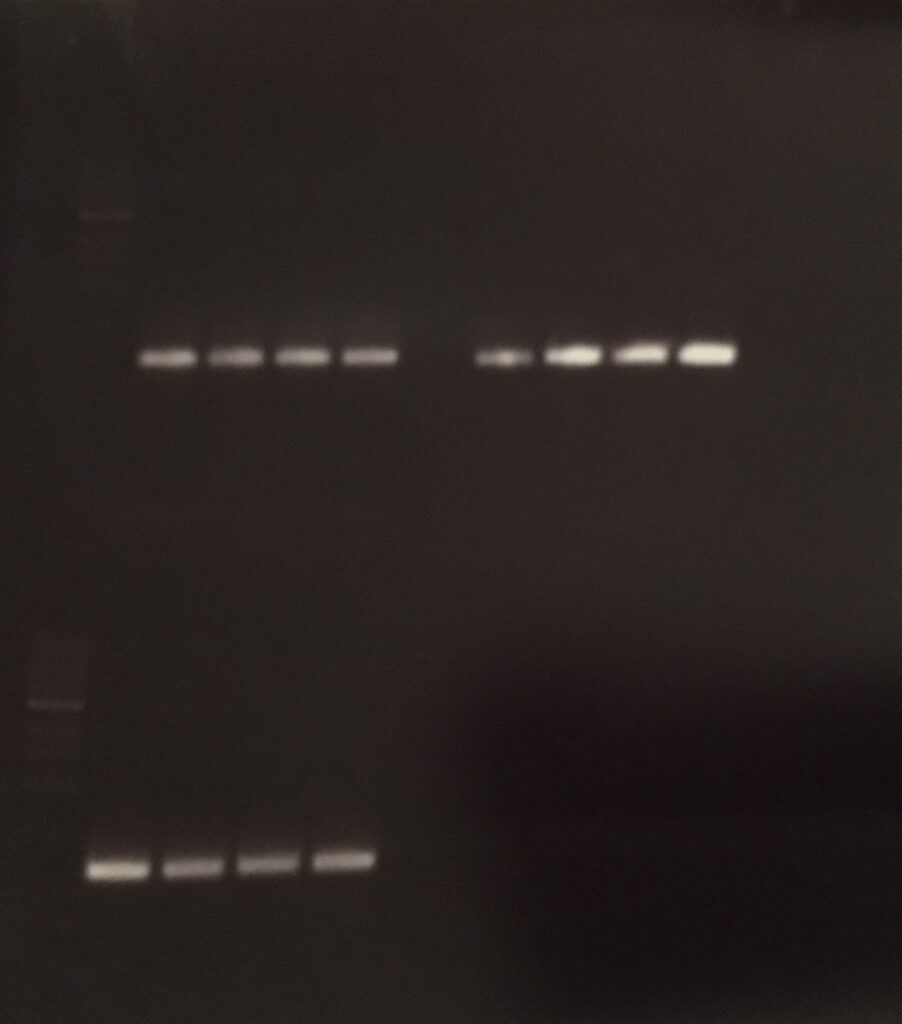
primer 1: 170.7 ng/µl
primer 2: 137.4 ng/µl
primer 3: 215.0 ng/µl
pCDF6 NEBuilder Assembly Reaction
-pcr1: 4.85 ng -> 1.60 µl
-pcr2: 4.24 ng -> 1.20 µl
-pcr3: 4.87 ng -> 1.20 µl
-pCDF6: 100 ng -> 2.90 µl
-H2O: 3.1 µl
-10 µl NEBuilder HiFi DNA Assembly Master Mix/Control
heat shock transformation
-> plated on LB0+Amp plates
-> no colonies
colony PCR (29.10.20)
-37 µl primer forward/reverse
-37 µl dNTPs
-37 µl Taq Polymerase
-74 µl buffer LSB
-518 µl H2O
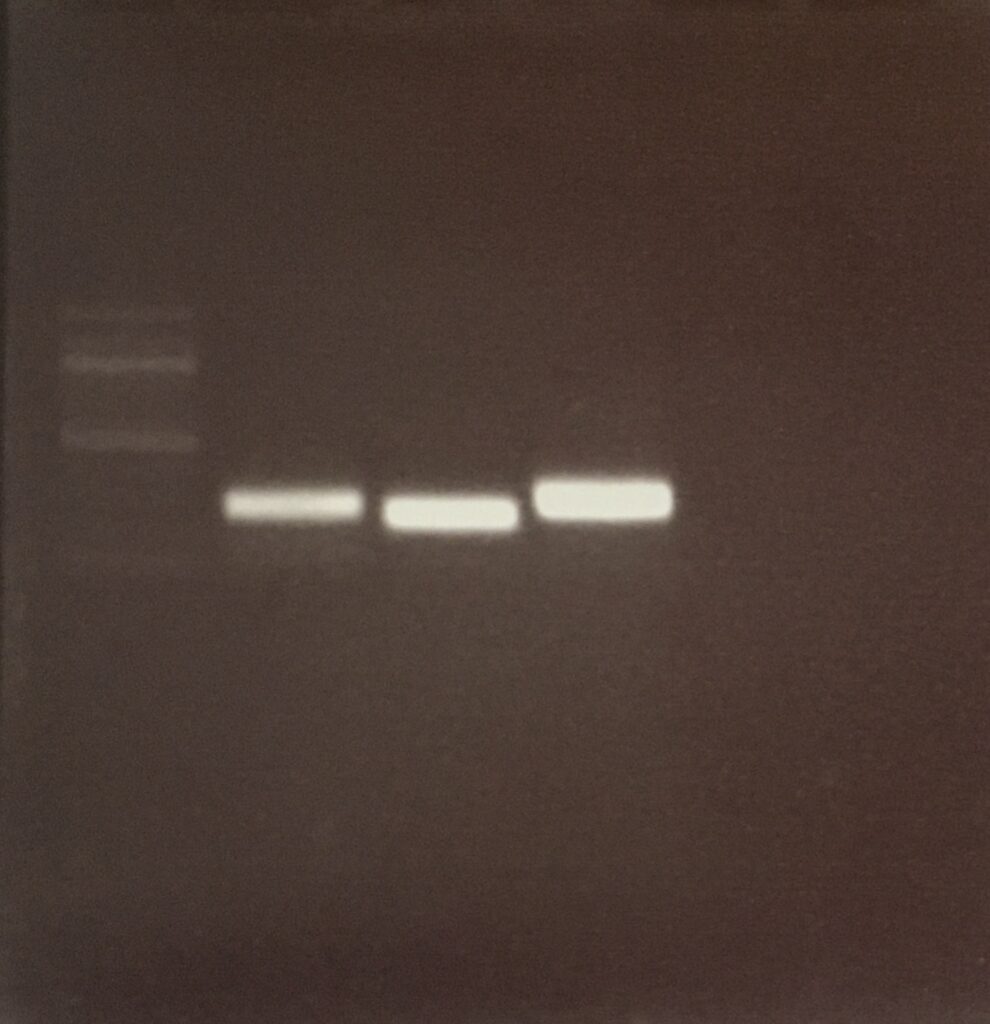
Agarose-gel electrophoresis
colony pcr