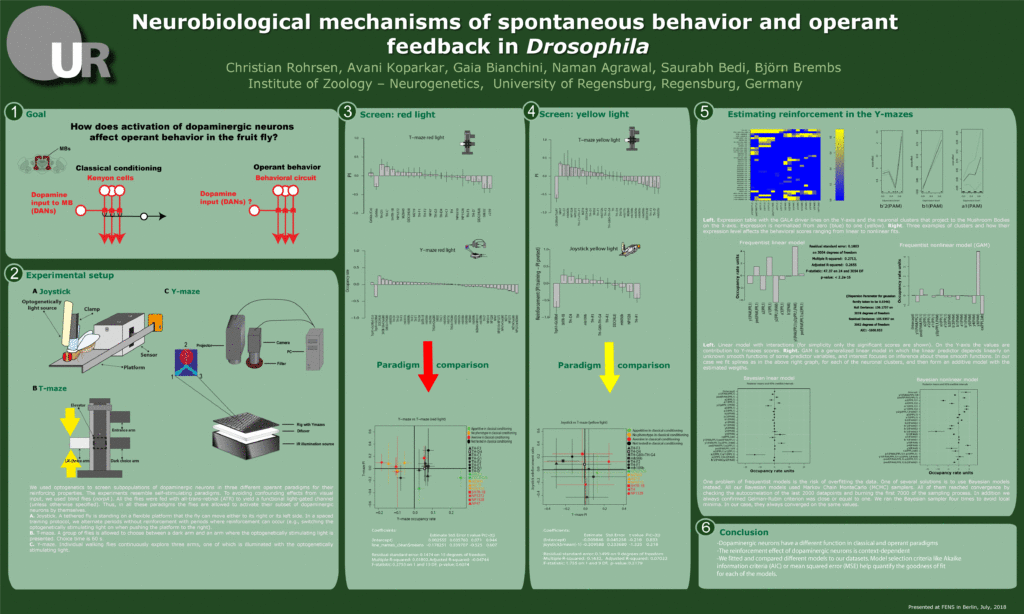
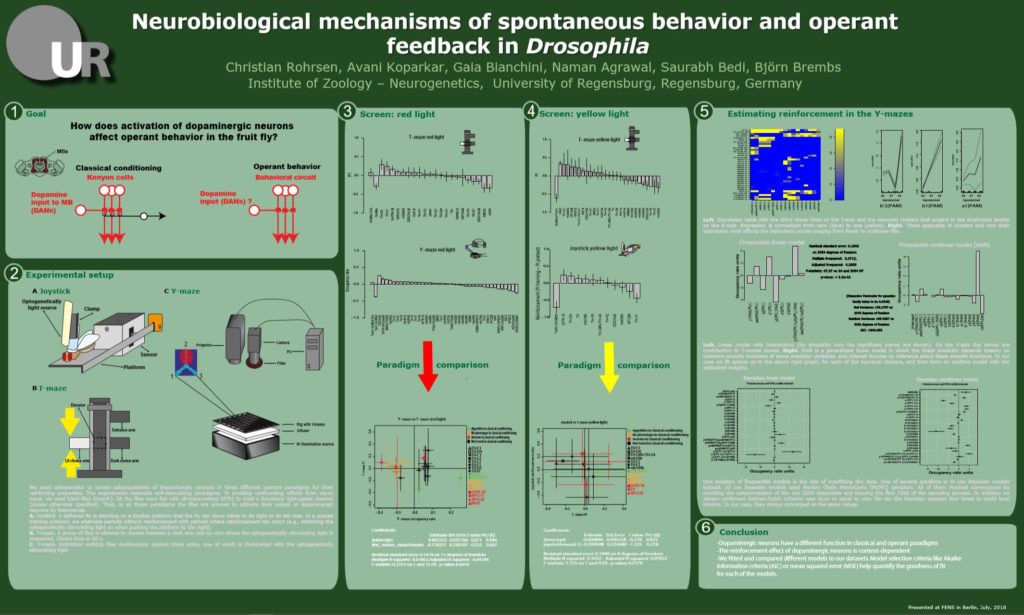
FENS poster (link for students)
on Thursday, February 14th, 2019 11:21 | by Christian Rohrsen
Category: Uncategorized | No Comments
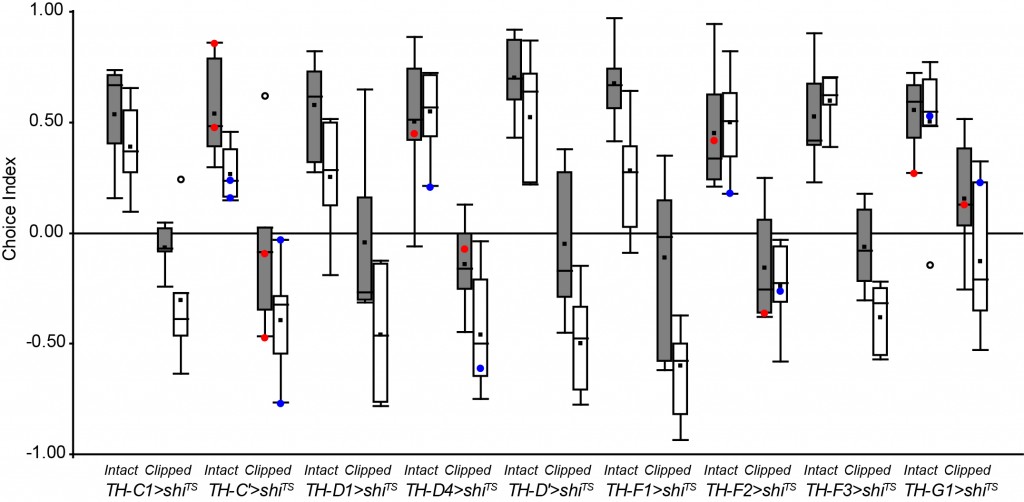
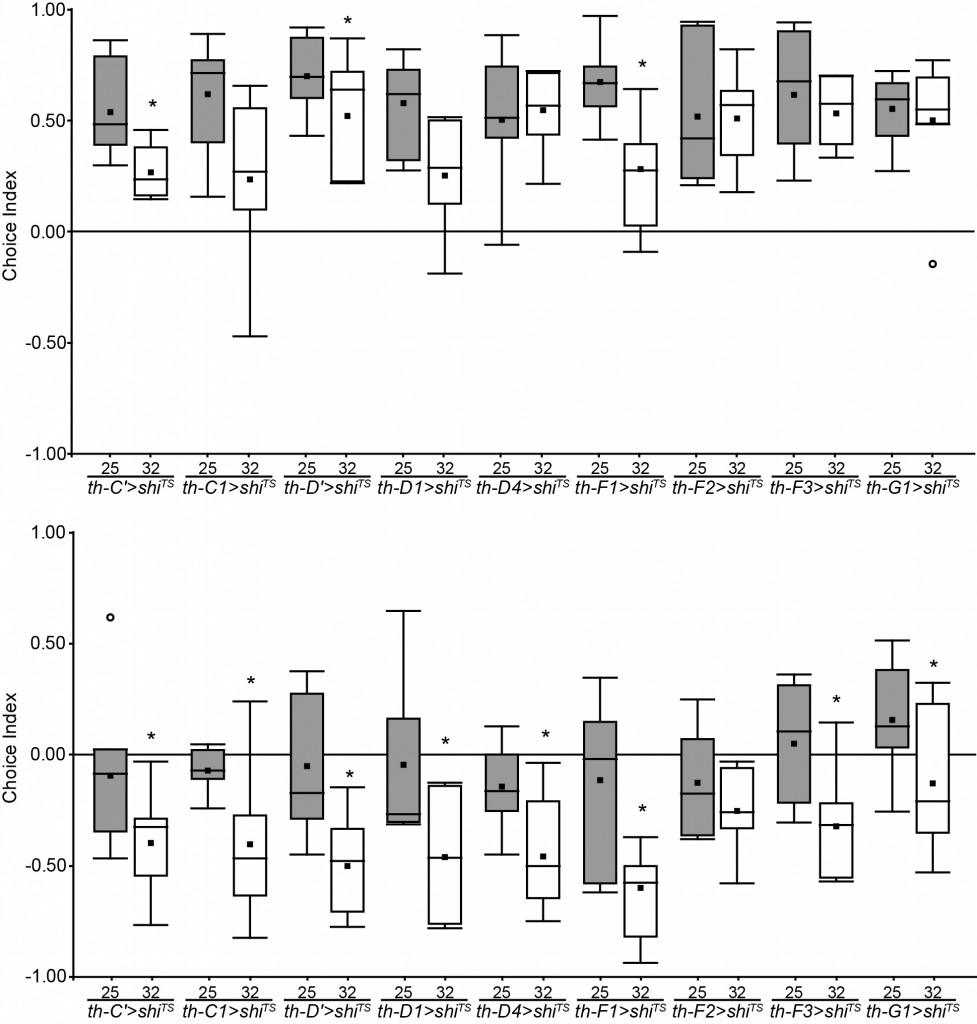
Update: Looking for the DA & OA neurons involved in phototactic flexibility
on Wednesday, October 7th, 2015 11:35 | by Axel Gorostiza
These are the newst results of my screen. There are some interesting candidates, but it’s still too soon to conclued something.
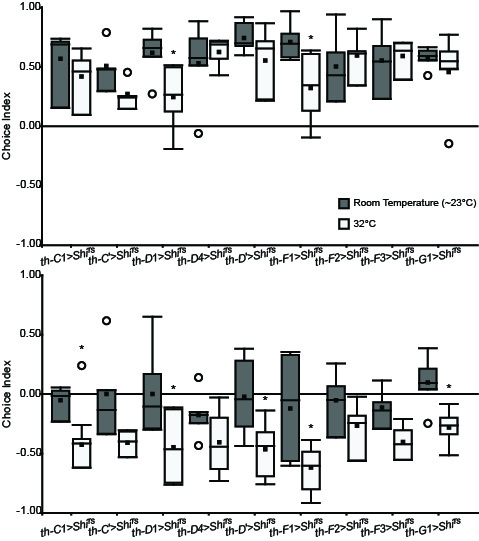
Update of the DA screening. New experiments are in red (25°C) and blue (32°C).
Experiment finished.
Category: wing clipping | No Comments