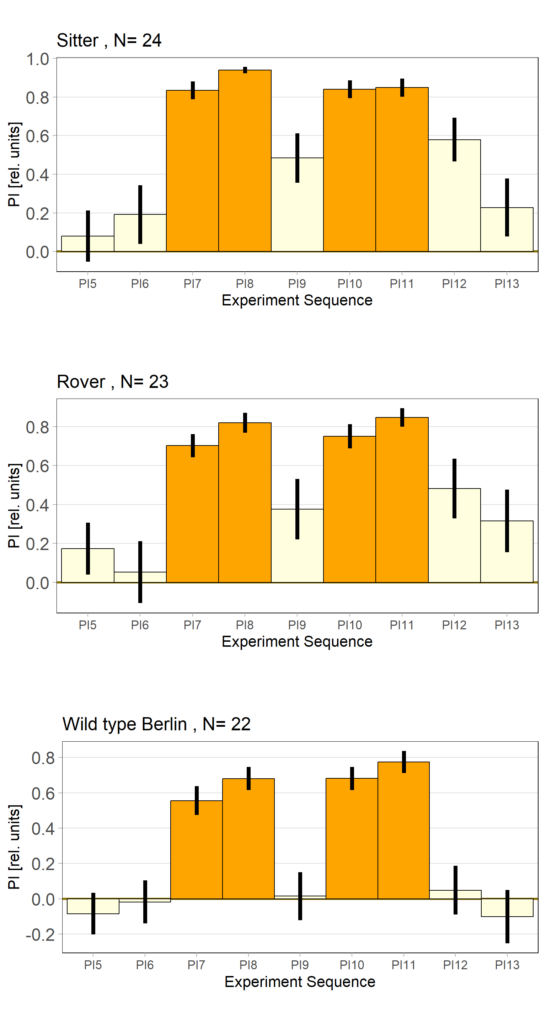
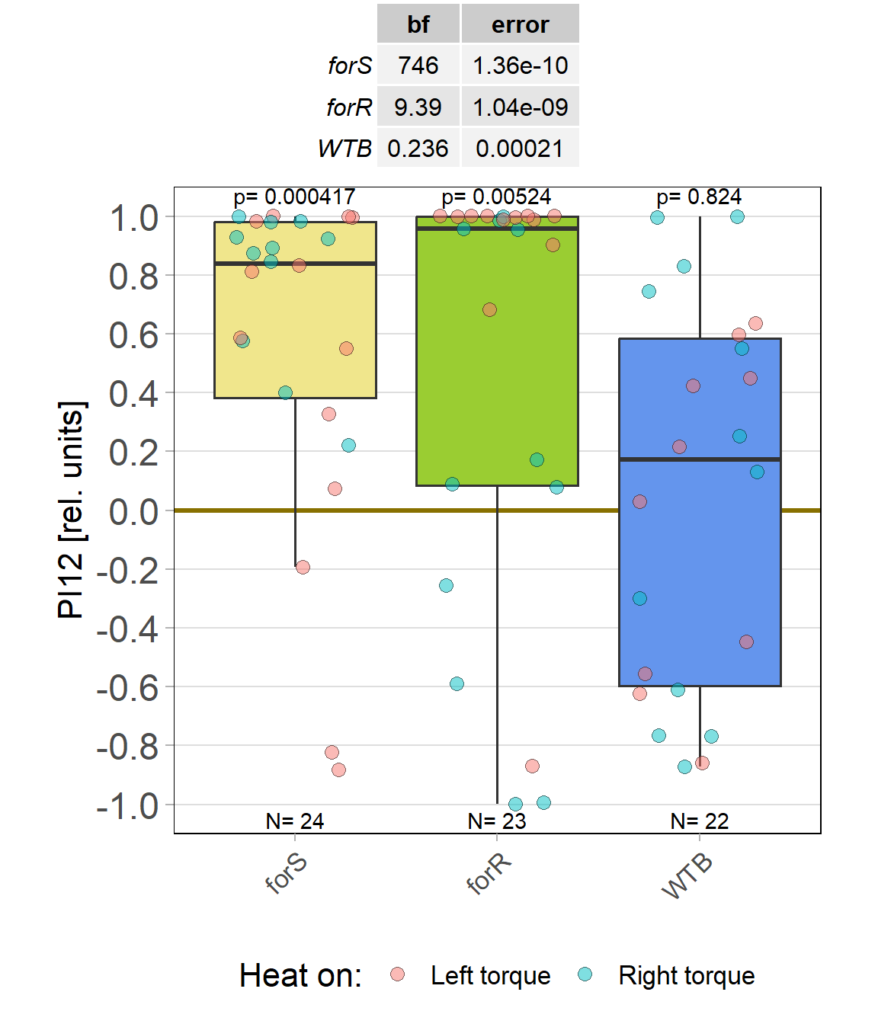
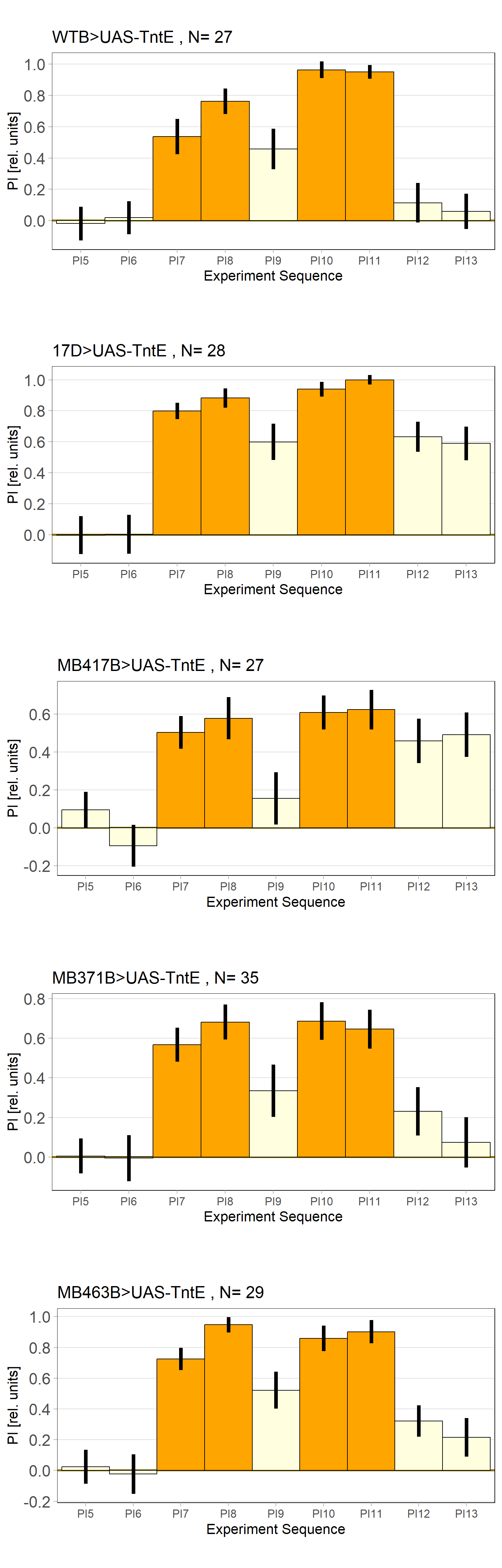
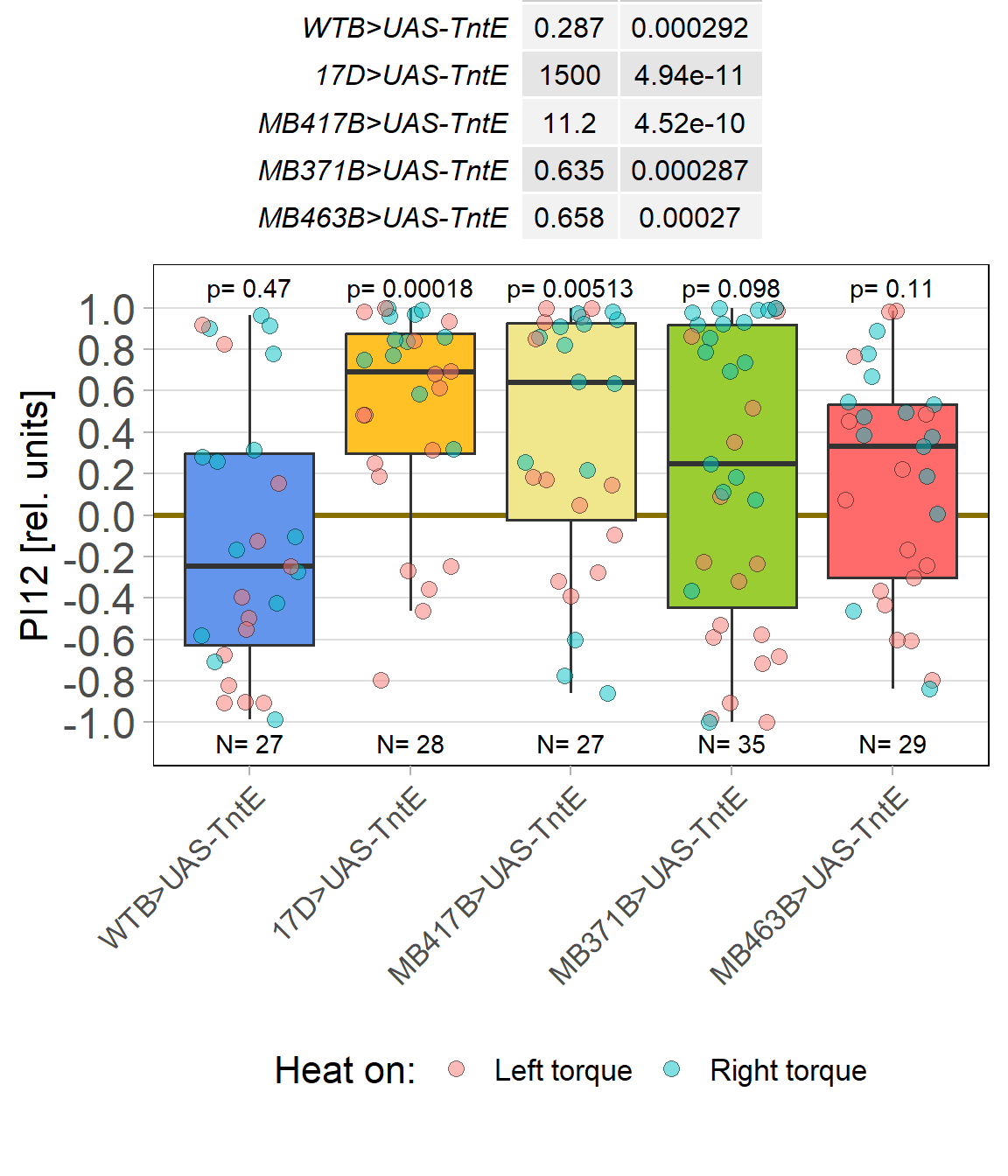
Rover vs. Sitter self learning after 4 minutes training
on Monday, October 16th, 2023 10:39 | by Radostina Lyutova
Category: flight, Habit formation, Memory, Operant learning, operant self-learning, Rover/Sitter | No Comments
Bachelor Blog / #7 offspring
on Monday, October 2nd, 2023 10:51 | by Ellie
Below you can find the data I collected from the offspring flies:
-> offspring from trained parents
-> offspring from untrained parents
(I left out data from flies that showed negative preference during two training periods in a row)
Bachelor Blog / #5 no learning :(
on Monday, September 11th, 2023 12:21 | by Ellie
Below you find the data from my experimental rounds A and B:
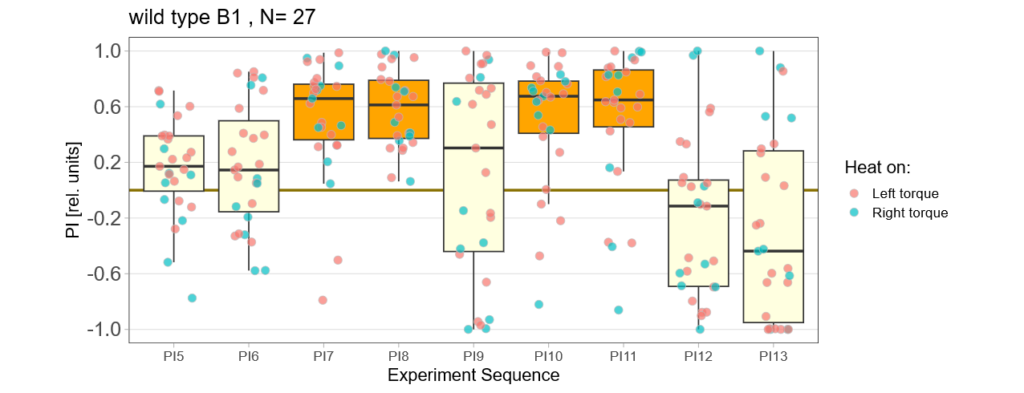
-> learning scores of the parental flies from experimental round A and B
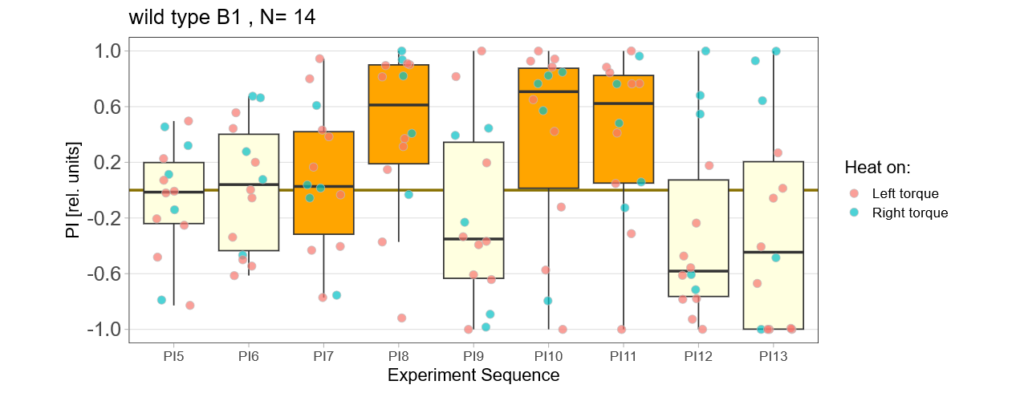
-> learning scores of the trained parent´s offspring only from experimental round A
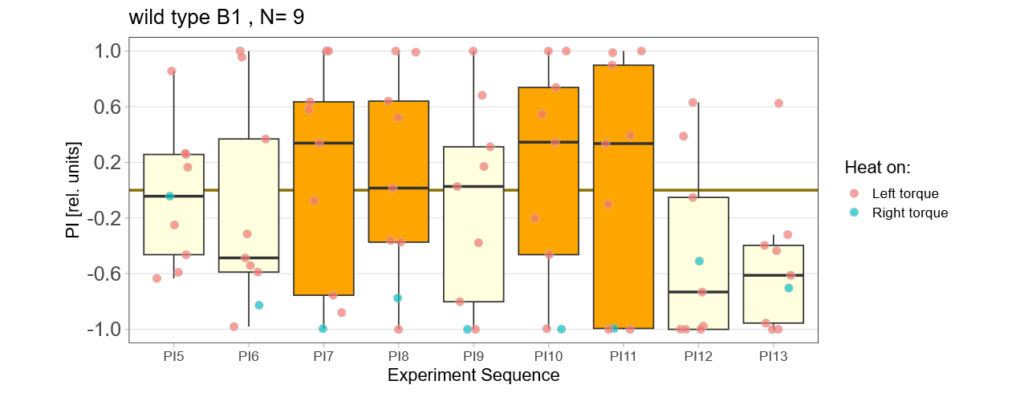
-> learning scores of the untrained parent´s offspring only from experimental round A
The results confuse me a lot and I am happy to discuss reasons :) However the offspring of the round-B will be ready for testing by the end of this week so there is still some data to collect…
Bachelor Blog / #4 is there something?
on Monday, August 7th, 2023 2:13 | by Ellie
The offspring of my first experimental fly cohort finally hatched! Below you find a few first pre-tests I ran last week :)
First, here are the results of a quick test to see if the offspring shows a preference for the parentally trained side after the first training period:
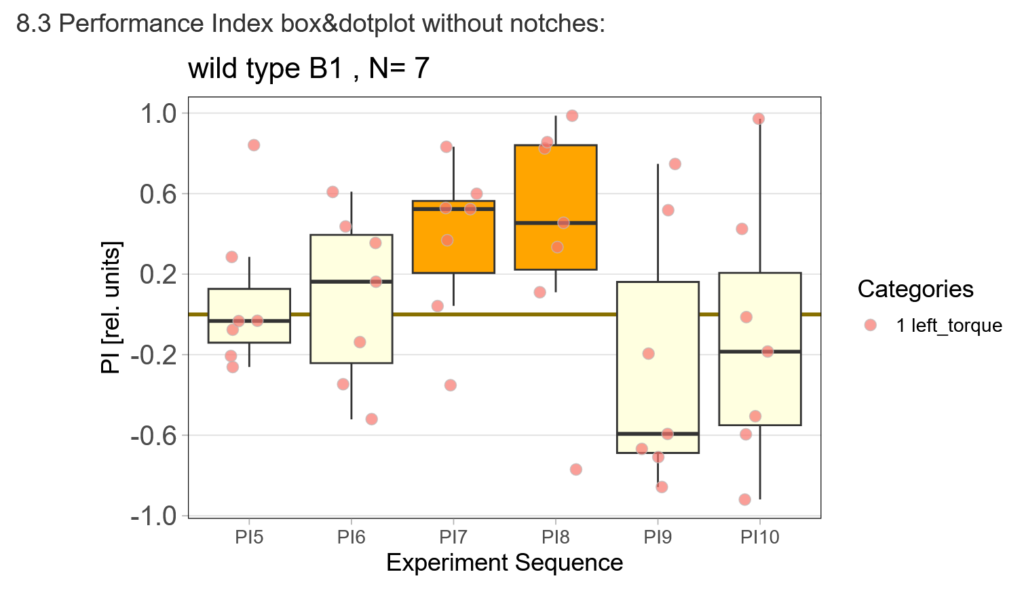
After that I played around with the laser a little bit to find the learning threshhold. I set the laser on 2,6V but the results I got look a bit weird:
-> untrained wtb flies
-> offspring of trained wtb flies
I´m optimistic however there is still a lot of work to do…
Inhibited subset of KCs in switch mode/yaw torque paradigm
on Friday, May 5th, 2023 3:52 | by Silvia Marcato
Category: flight, Habit formation, Kenyon cells, Mushroom Body | No Comments
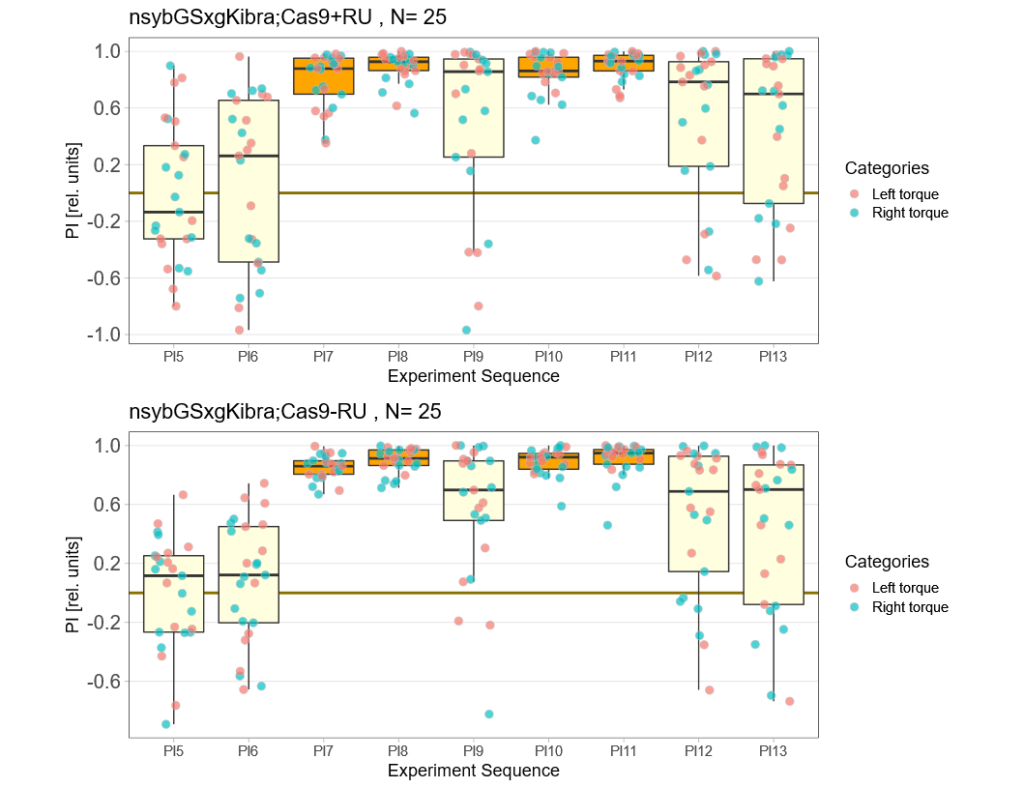
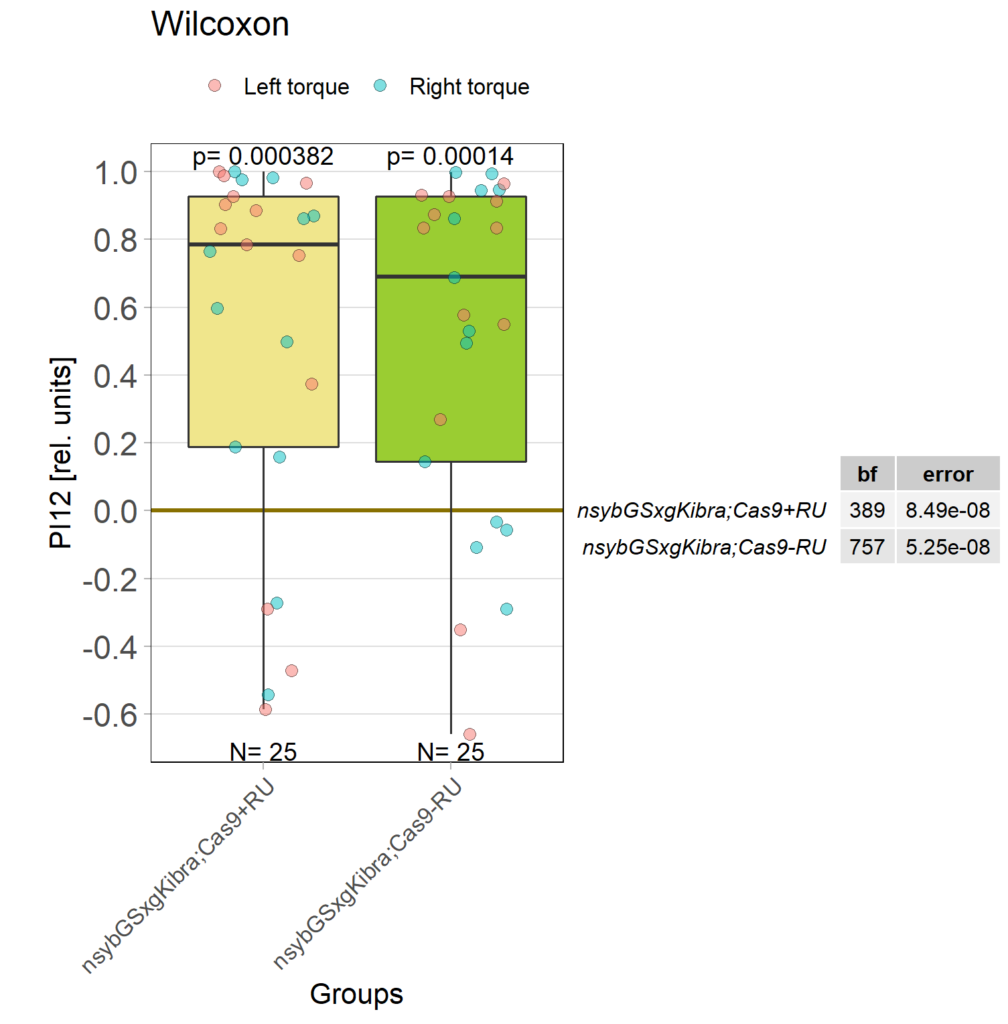
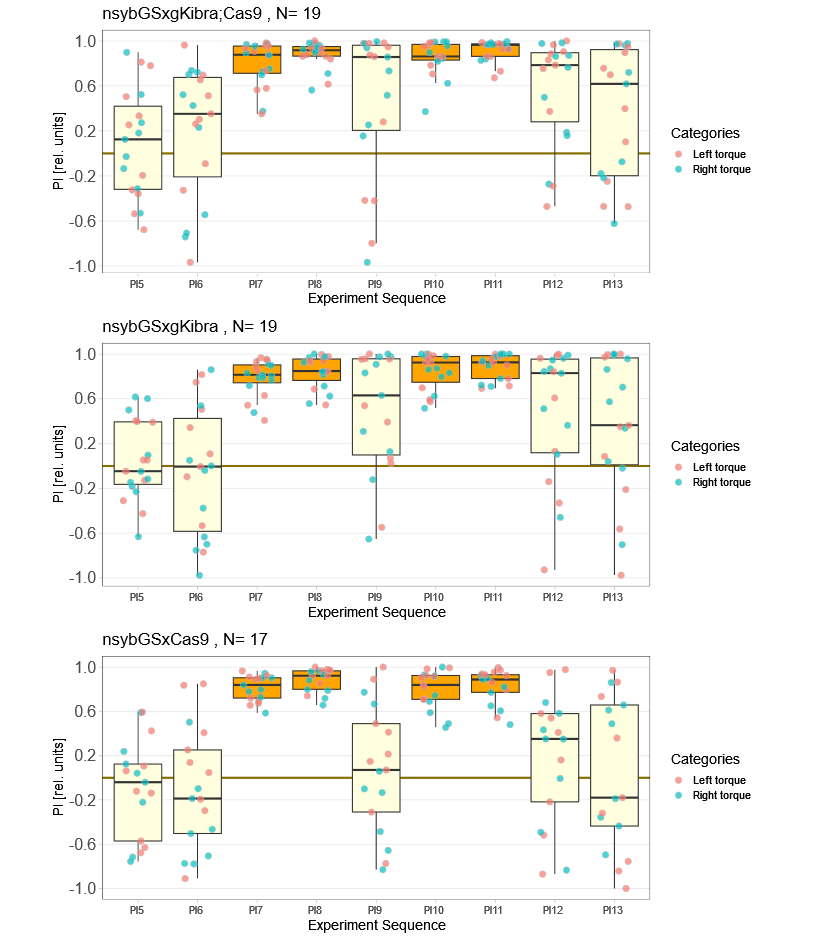
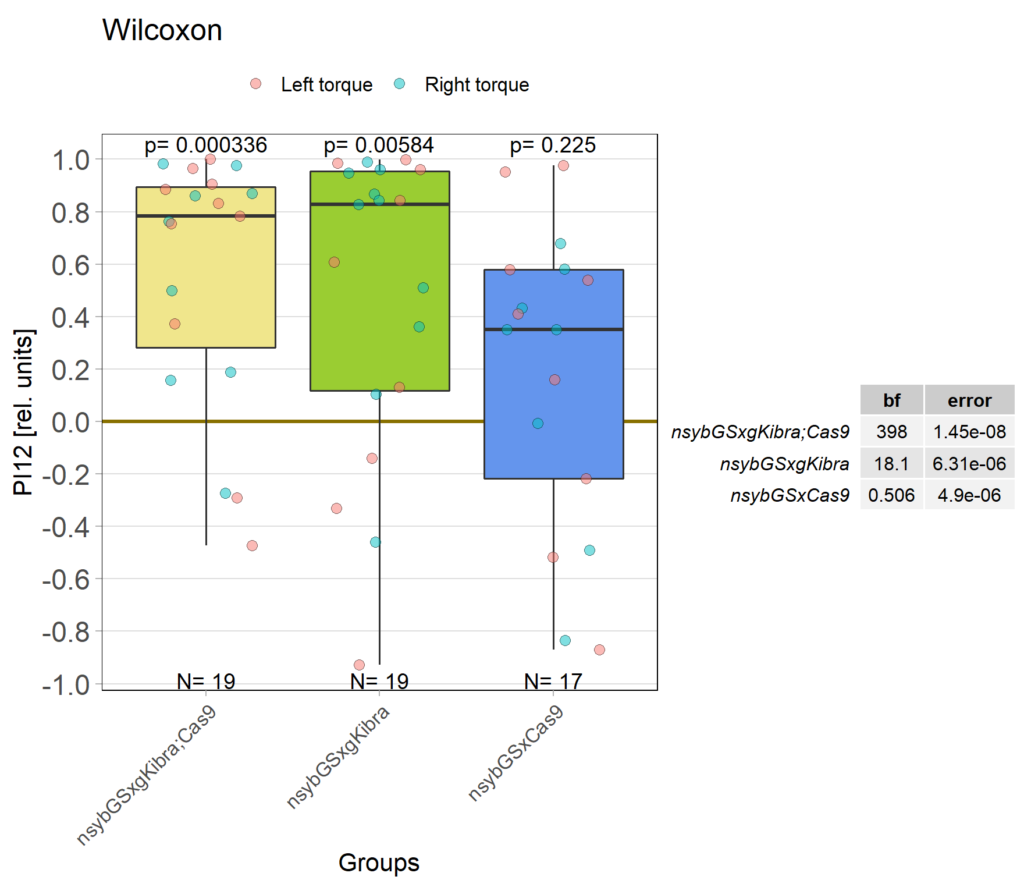
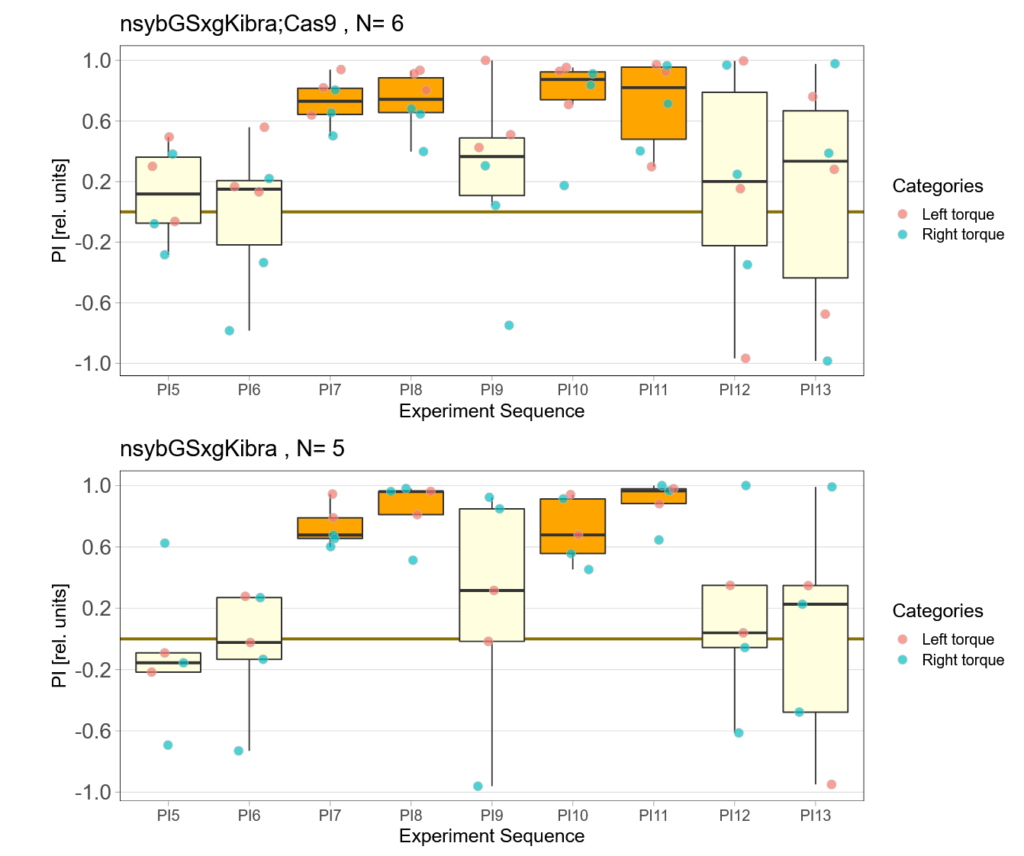
Kibra knockout, adult
on Monday, December 19th, 2022 11:15 | by Andreas Ehweiner
Category: flight, Memory, Operant learning, operant self-learning, Uncategorized | No Comments
Kibra knockout, adult
on Monday, November 28th, 2022 11:47 | by Andreas Ehweiner
Category: flight, Operant learning, operant self-learning | No Comments
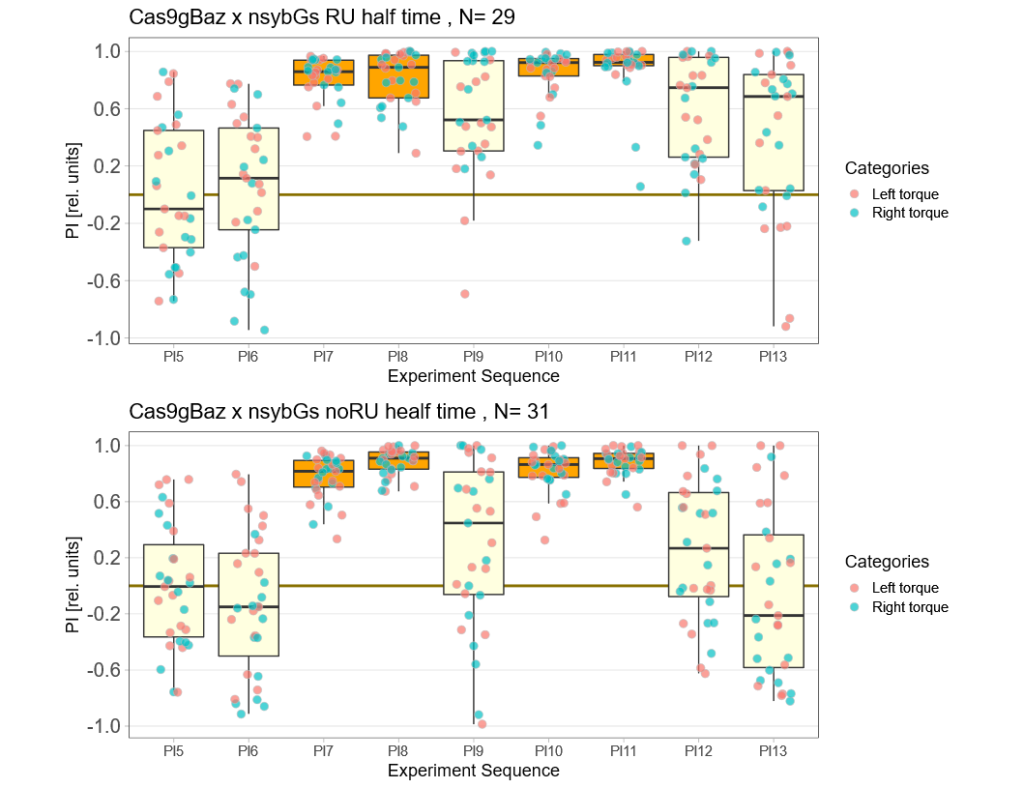
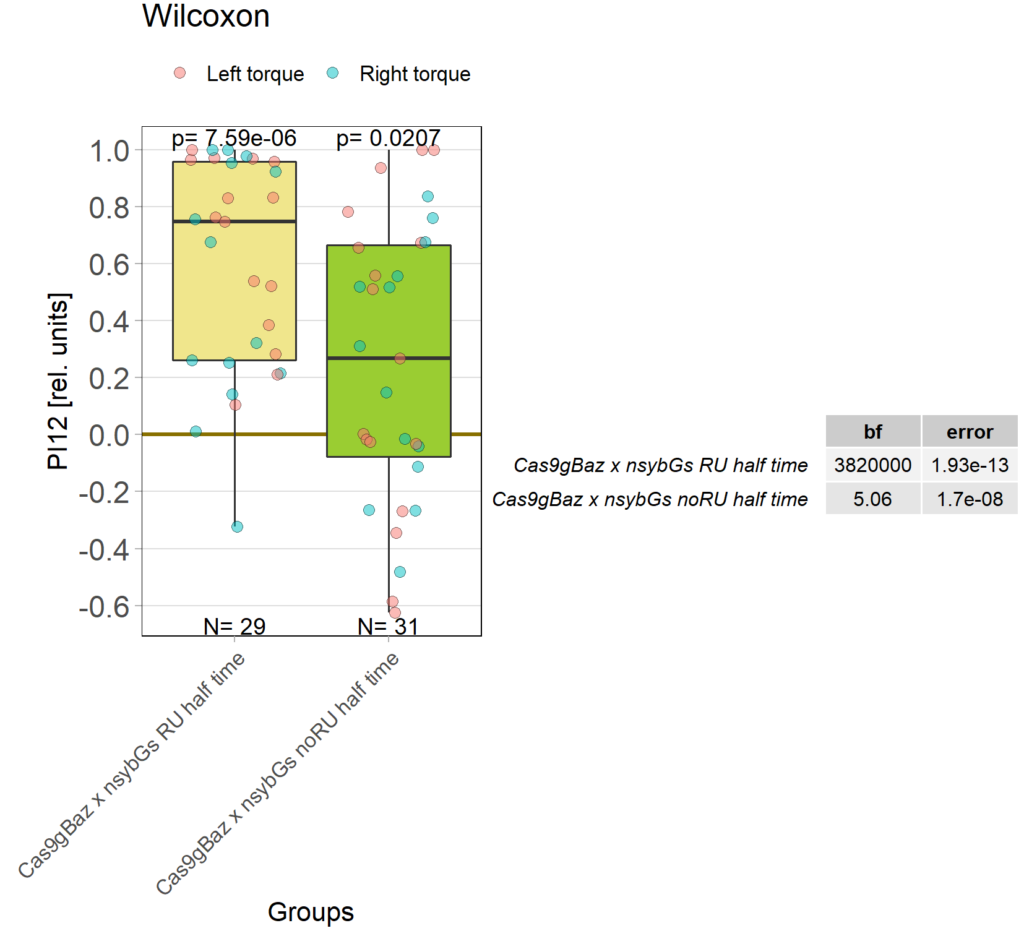
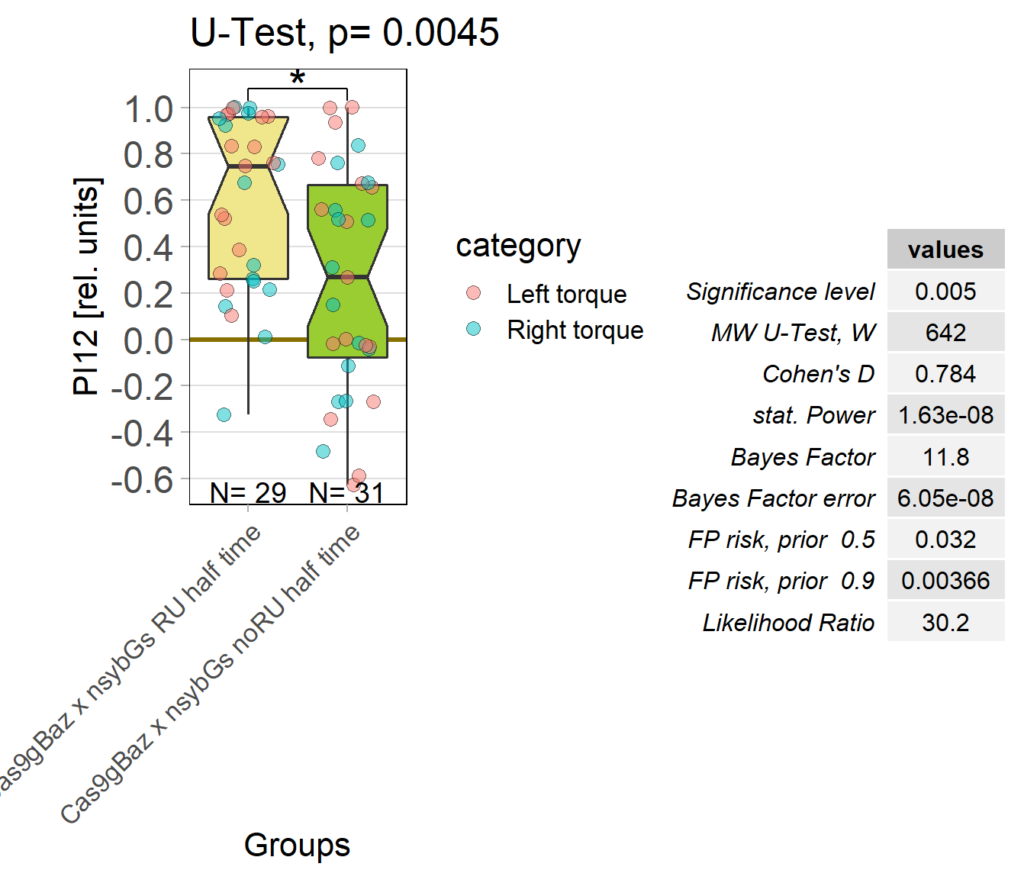
baz adult knockout, half time
on Monday, October 17th, 2022 1:08 | by Andreas Ehweiner
Category: flight, Operant learning, operant self-learning | No Comments
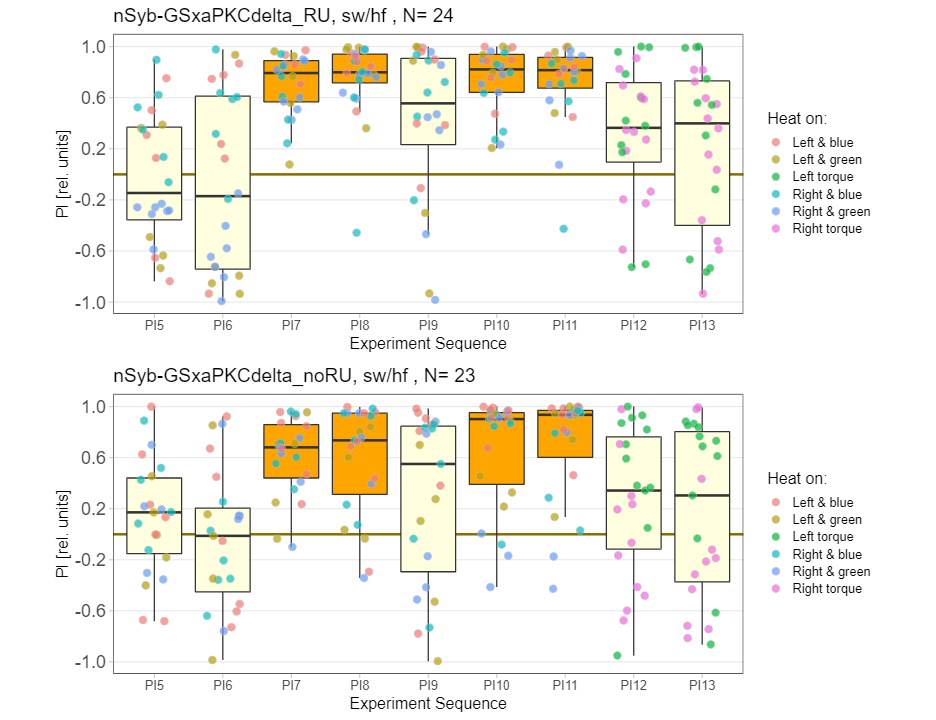
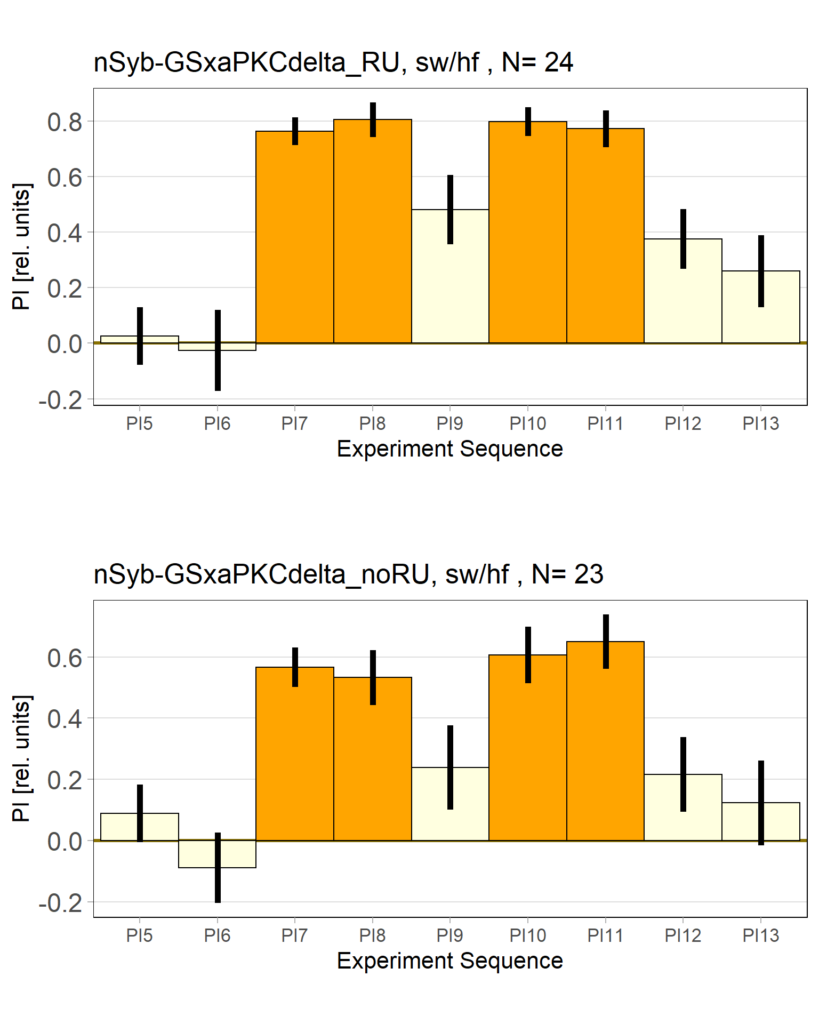
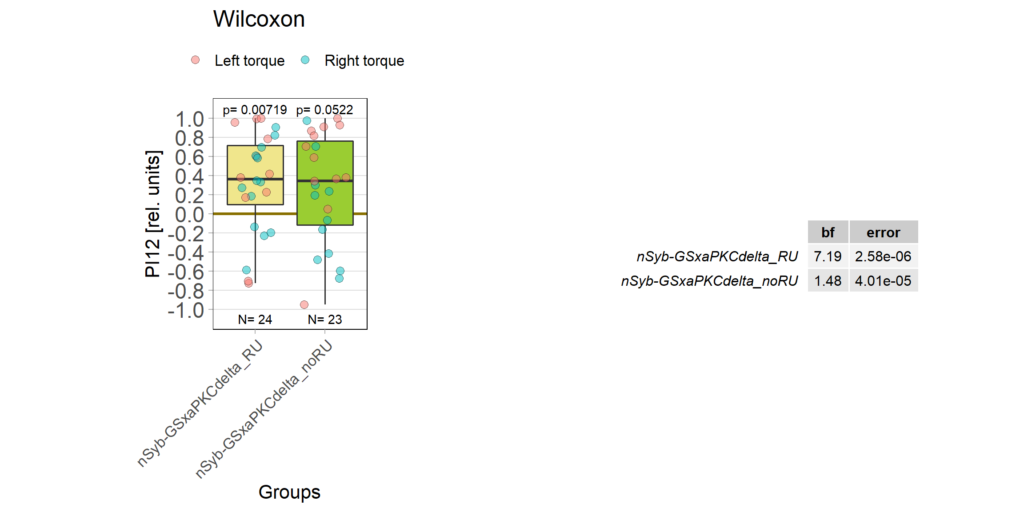
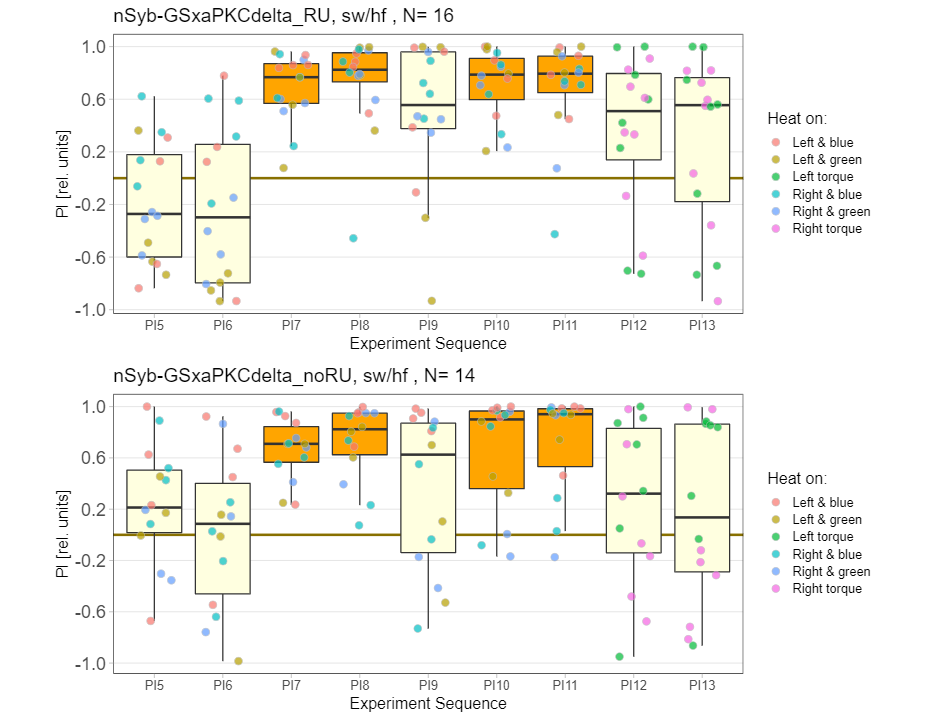
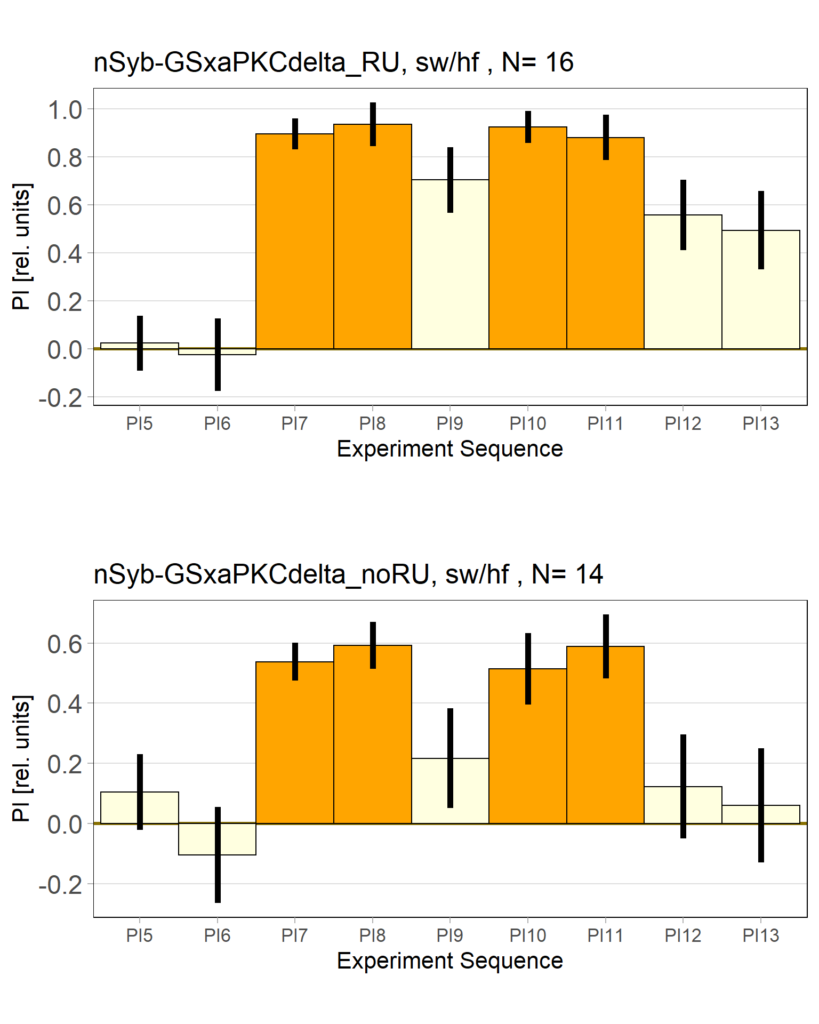
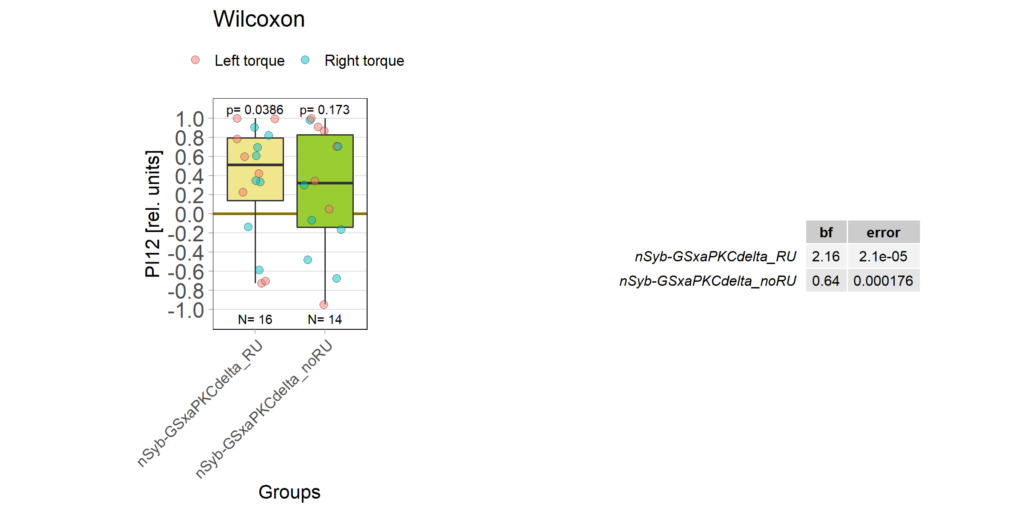
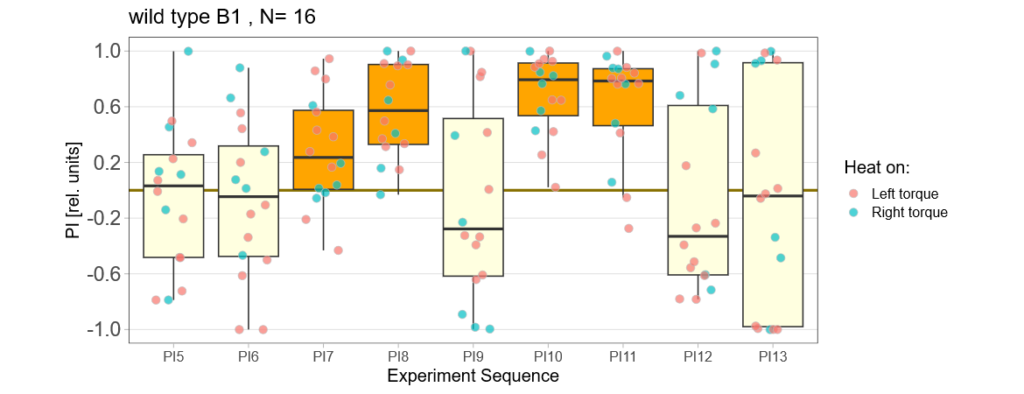
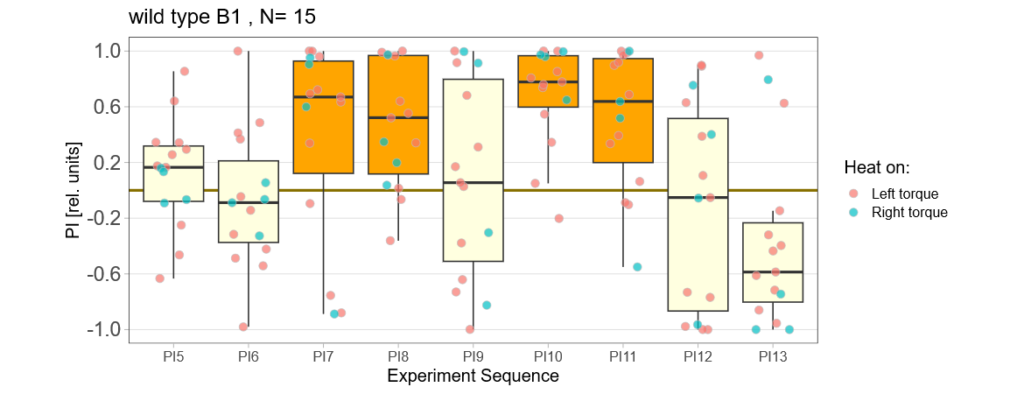
panneuronal aPKC overexpression in switch mode/yaw torque paradigm
on Monday, October 10th, 2022 10:06 | by Radostina Lyutova
Category: flight, Habit formation, Memory, Operant learning, operant self-learning, PKC, World learning | No Comments
panneuronal aPKC overexpression in switch mode/yaw torque paradigm
on Tuesday, October 4th, 2022 1:50 | by Radostina Lyutova
Category: flight, Habit formation, Operant learning, operant self-learning, PKC, World learning | No Comments